Irb

The Institutional Review Board Irb Kareem Coomansingh Msc Mph Irb An institutional review board (irb) is a committee that reviews research involving human subjects to ensure ethical standards. learn about the history, purpose, regulations, and exceptions of irbs in the united states and other countries. Although institutions engaged in research involving human subjects will usually have their own irbs to oversee research conducted within the institution or by the staff of the institution, fda.

Irb Organizational Chart Gcgmh The institutional review board (irb) was established to assist researchers in the protection of the rights and welfare of human subjects involved in research. These entities are often called institutional review boards (irb), ethical review boards (erb), independent ethics committees (iec), or research ethics boards (rec) (usaid, 2018). the institutional review board (irb) is a research ethics committee that reviews and approves human subjects’ research. Learn how to register and update your institutional review board (irb) with ohrp, the federal agency that oversees human subjects research. find faqs, tutorials and links to other resources on irb registration. The irb serves to protect the rights and welfare of research study participants.

Irb Organizational Chart Gcgmh Learn how to register and update your institutional review board (irb) with ohrp, the federal agency that oversees human subjects research. find faqs, tutorials and links to other resources on irb registration. The irb serves to protect the rights and welfare of research study participants. The institutional review board is charged with the responsibility of reviewing, prior to its initiation, research involving human participants. the irb is primarily concerned with ensuring that the rights, welfare, and privacy of human participants are protected. The irb has the authority to approve, require modifications in, or disapprove all research activities that fall within its jurisdiction as specified in the federal regulations, state regulations and statutes, and institutional policy. Ensure irb compliance across every study navigating irb requirements demands precision, documentation discipline, and a proactive approach to compliance. whether you are submitting a new protocol, responding to a modification request, or preparing for an audit, maintaining clear and complete records at every stage is critical to protecting your study timelines and outcomes. wcg’s ereview. Institutions seeking federal funding must have an irb, and the irb must review and approve federally funded research studies. most institutions require irb approval for all research that involves human subjects, not only that funded by the federal government.
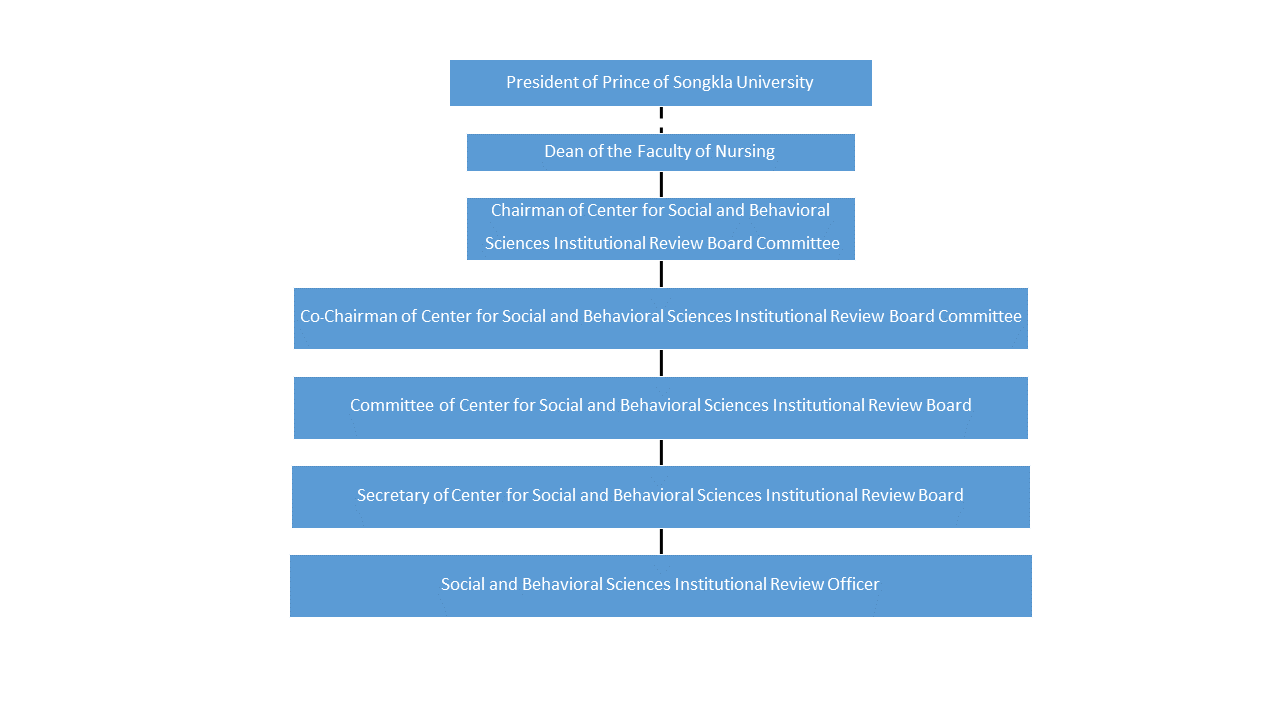
Institutional Review Board Irb Psu The institutional review board is charged with the responsibility of reviewing, prior to its initiation, research involving human participants. the irb is primarily concerned with ensuring that the rights, welfare, and privacy of human participants are protected. The irb has the authority to approve, require modifications in, or disapprove all research activities that fall within its jurisdiction as specified in the federal regulations, state regulations and statutes, and institutional policy. Ensure irb compliance across every study navigating irb requirements demands precision, documentation discipline, and a proactive approach to compliance. whether you are submitting a new protocol, responding to a modification request, or preparing for an audit, maintaining clear and complete records at every stage is critical to protecting your study timelines and outcomes. wcg’s ereview. Institutions seeking federal funding must have an irb, and the irb must review and approve federally funded research studies. most institutions require irb approval for all research that involves human subjects, not only that funded by the federal government.
Comments are closed.