What Needs Irb Review Navigating Rules And Processes

Navigating The Irb While Conducting Research In Your Teaching Practice This guidance document is consistent with the goals of section 3023 of the cures act. this guidance is intended for institutions and institutional review boards (irbs) responsible for review and oversight of human subject research under the hhs and fda regulations. Studies generally need at least an annual irb review. in addition, study modifications (i.e., changes to recruitment plans), wherein the investigator changes an aspect of an already approved irb protocol, require another review by the irb.
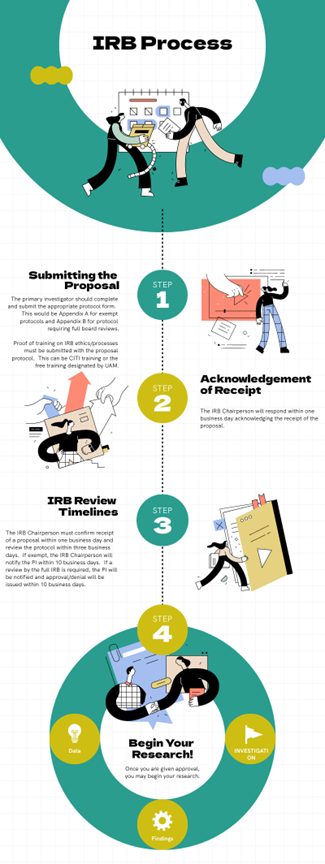
Irb Review Process Irb information sheet frequently asked questions i. irb organization 1. what is an institutional review board (irb)? under fda regulations, an irb is an appropriately constituted group that has. Compiled by the uab office of the institutional review board (oirb), it highlights key considerations for ensuring ethical compliance, regulatory adherence, and submission success. this guidebook provides researchers with clear directions to navigate the irb submission process. The following guidance will help you evaluate when the nu irb does not need to be consulted regarding a “fee for service” activity that collects, accesses, uses, or analyzes human subjects data or biospecimens (regardless of identifiability). Institutional review boards (irbs) are federally required committees that evaluate research involving human subjects to safeguard participants’ rights, safety, and well being. their oversight guarantees adherence to federal laws, institutional policies, and ethical standards.

Irb Review Process Research Protections The following guidance will help you evaluate when the nu irb does not need to be consulted regarding a “fee for service” activity that collects, accesses, uses, or analyzes human subjects data or biospecimens (regardless of identifiability). Institutional review boards (irbs) are federally required committees that evaluate research involving human subjects to safeguard participants’ rights, safety, and well being. their oversight guarantees adherence to federal laws, institutional policies, and ethical standards. This section outlines the composition, operation, types of review, and criteria for irb approval, providing a roadmap for navigating the irb process effectively. Explore the critical roles and ethical responsibilities of institutional review boards (irbs) in clinical research, human subject protection, and compliance. This article breaks down the irb approval process step by step, offering essential insights that equip researchers to handle this pivotal part of their work efficiently. The irb reviews studies involving drugs, devices, biologics, radioactive materials, and in vitro diagnostic devices in accordance with relevant fda regulations.
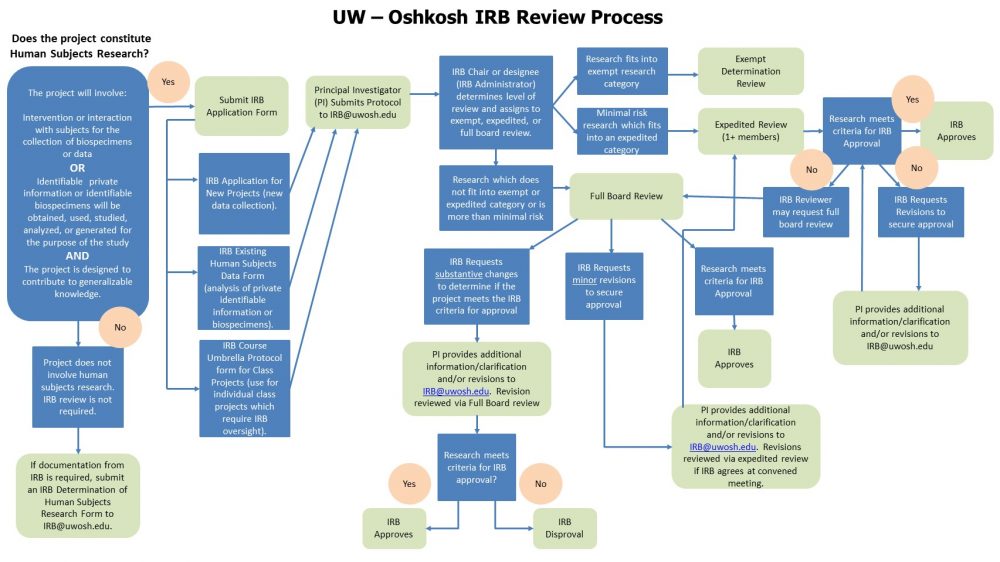
Irb Review Process Flowchart 2022 Office Of Sponsored Programs This section outlines the composition, operation, types of review, and criteria for irb approval, providing a roadmap for navigating the irb process effectively. Explore the critical roles and ethical responsibilities of institutional review boards (irbs) in clinical research, human subject protection, and compliance. This article breaks down the irb approval process step by step, offering essential insights that equip researchers to handle this pivotal part of their work efficiently. The irb reviews studies involving drugs, devices, biologics, radioactive materials, and in vitro diagnostic devices in accordance with relevant fda regulations.
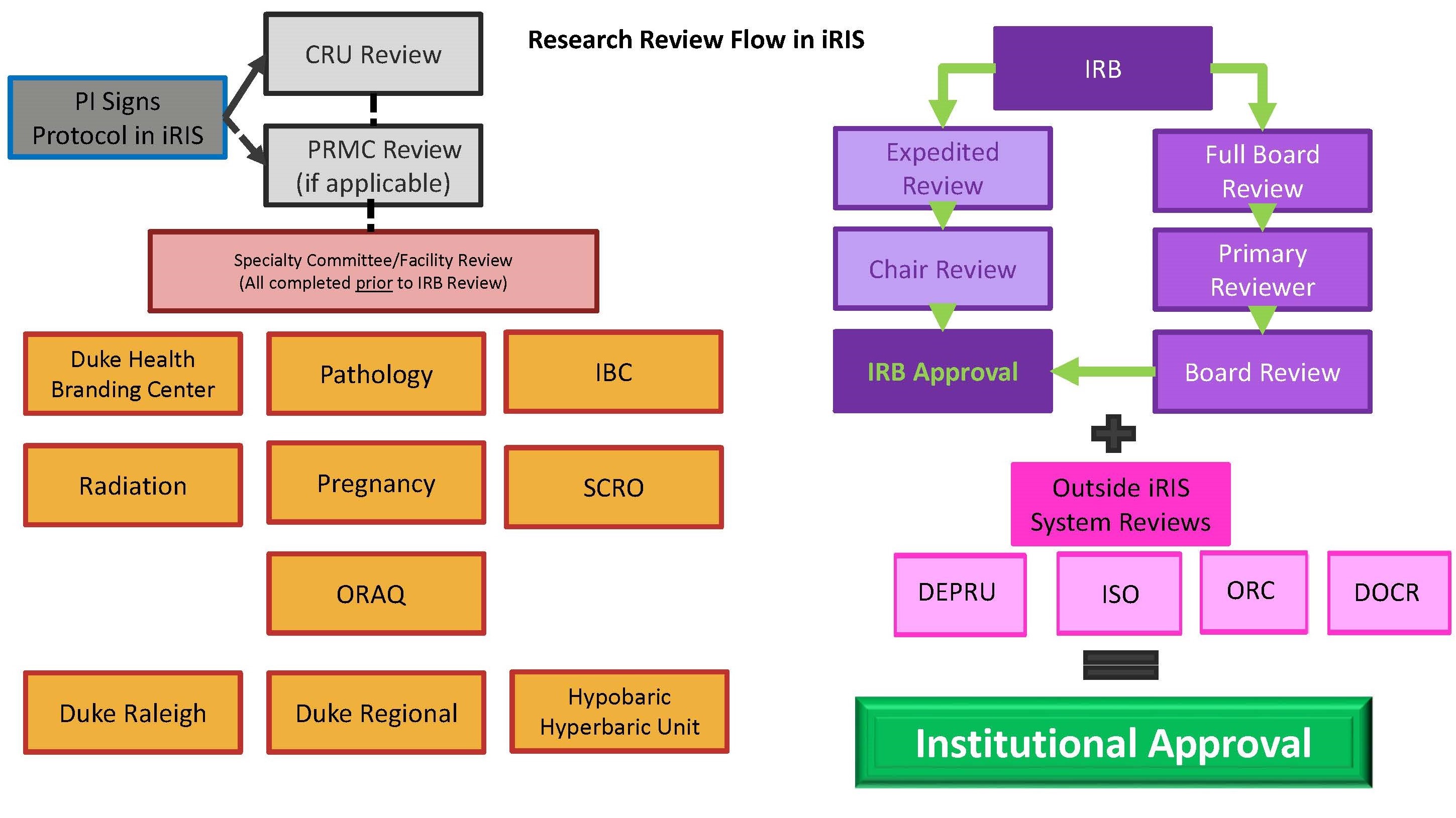
Irb Process Duke Health Institutional Review Board This article breaks down the irb approval process step by step, offering essential insights that equip researchers to handle this pivotal part of their work efficiently. The irb reviews studies involving drugs, devices, biologics, radioactive materials, and in vitro diagnostic devices in accordance with relevant fda regulations.
Comments are closed.