Institutional Review Board Irb Explained Ccrps

Institutional Review Board Irb Explained Ccrps What is an institutional review board (irb)? an institutional review board (irb) is a group of experts and professionals—such as doctors, scientists, ethicists, and legal advisors—that is responsible for reviewing and overseeing research studies involving human participants. The institutional review board (irb) is a research ethics committee that reviews and approves research involving human subjects. the irb’s primary responsibility is to protect the rights and welfare of research participants.

Institutional Review Board Irb Explained Ccrps Explore the critical roles and ethical responsibilities of institutional review boards (irbs) in clinical research, human subject protection, and compliance. This guidance document is consistent with the goals of section 3023 of the cures act. this guidance is intended for institutions and institutional review boards (irbs) responsible for review and oversight of human subject research under the hhs and fda regulations. Institutional review boards (irbs) are federally required committees that evaluate research involving human subjects to safeguard participants’ rights, safety, and well being. their oversight guarantees adherence to federal laws, institutional policies, and ethical standards. This lesson will explain the purpose and membership requirements of institutional review boards, or irbs. this lesson focuses on the revised common rule (or 2018 requirements) that became effective in 2018.
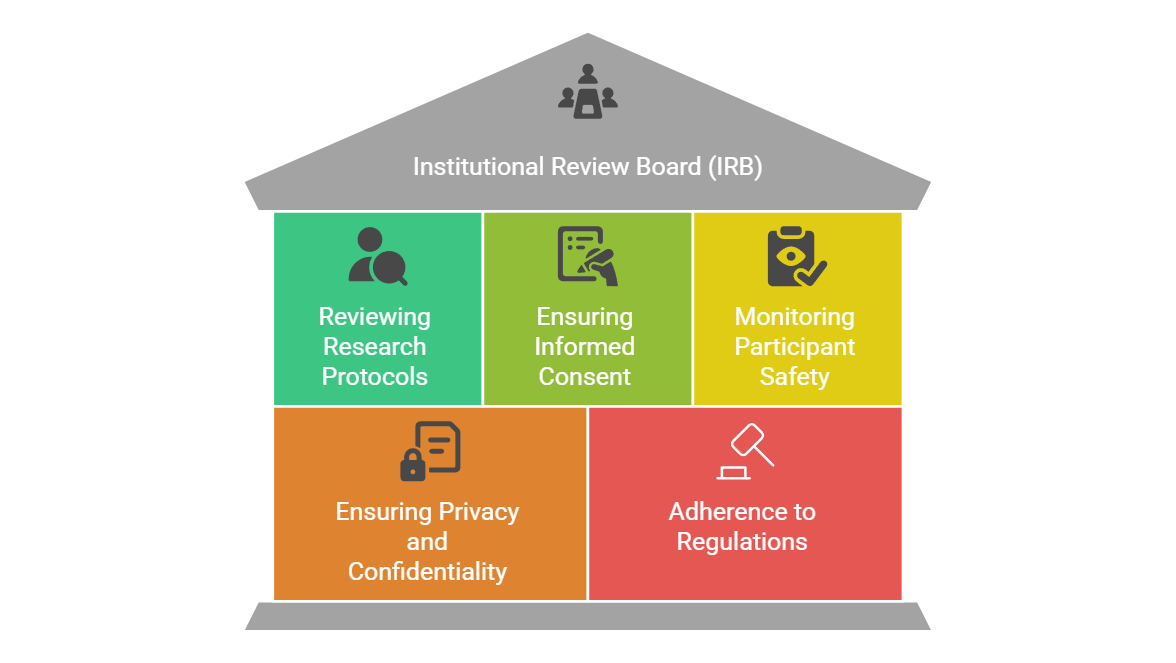
Institutional Review Board Irb Explained Ccrps Institutional review boards (irbs) are federally required committees that evaluate research involving human subjects to safeguard participants’ rights, safety, and well being. their oversight guarantees adherence to federal laws, institutional policies, and ethical standards. This lesson will explain the purpose and membership requirements of institutional review boards, or irbs. this lesson focuses on the revised common rule (or 2018 requirements) that became effective in 2018. The institutional review board (irb) is a committee charged with the responsibility of reviewing and overseeing all research proposals that involve human subjects. the board is comprised of scientists, non scientists, and community members. Understand what an irb is, what it does, and how it works. learn about the federal and regulatory guidelines, timelines, and history of institutional review boards. Each institutional review board (irb) that reviews research involving human subjects conducted or supported by the department of health and human services (hhs) must be registered with the office for human research protections (ohrp). Explore the essential role of institutional review boards (irbs) in guiding and overseeing clinical research.
Comments are closed.