The Irb Process

Irb Process Pdf Institutional Review Board The irb notifies the investigator of the outcome of the review process. if the research is approved, the study may begin, and the consent form and recruitment materials will get an irb approval stamp. Federal regulation and institutional operating procedure require that the irb reviews all the research documents and activities that bear directly on the rights and welfare of the subjects of proposed research.
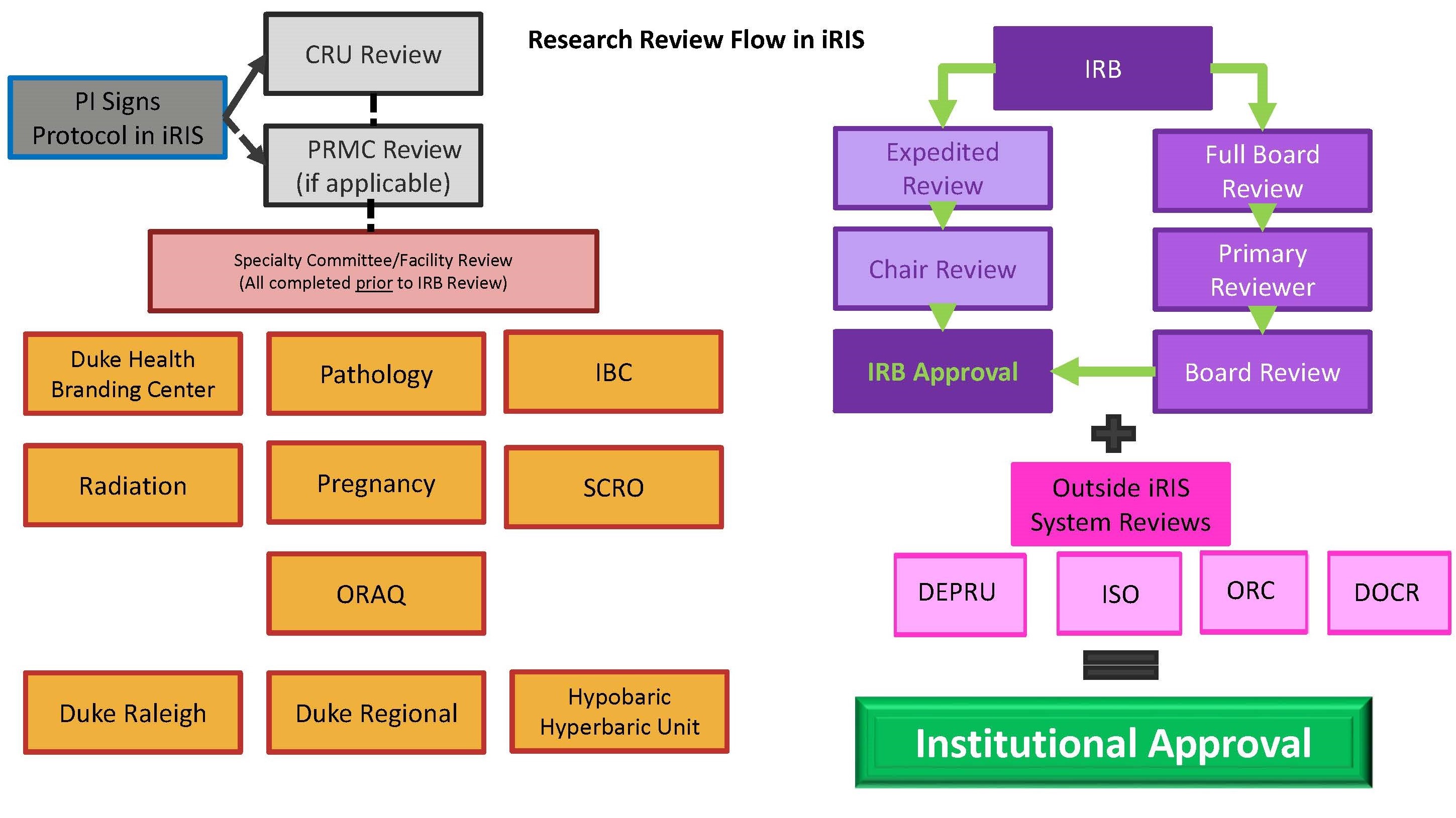
Irb Process Duke Health Institutional Review Board What is an institutional review board (irb)? under fda regulations, an irb is an appropriately constituted group that has been formally designated to review and monitor biomedical research. Institutions irbs may choose to create written procedures that focus solely on the regulatory responsibilities of the irb, or they may choose to also incorporate institutional policies and procedures that are a function of the institution’s human research protections program (hrpp). An irb’s core job is to protect the rights and welfare of people who volunteer for research. it does this in two phases: reviewing the study before it begins and monitoring it periodically while it’s underway. Irb processes are integral to every stage of a clinical trial, from protocol development and initial submission to amendments and final reporting. mastery of irb requirements ensures that ethical principles are built into study design from the ground up.
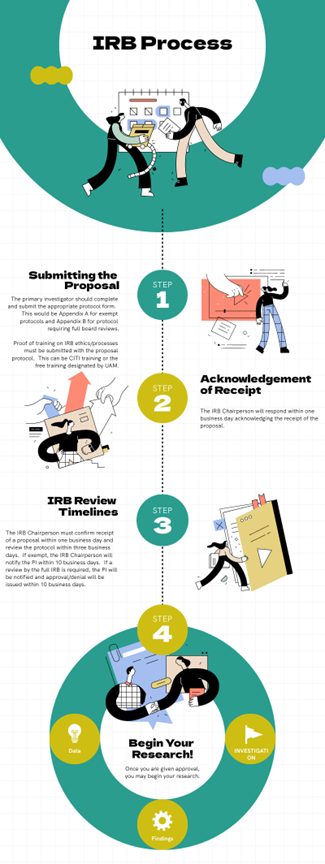
Irb Review Process An irb’s core job is to protect the rights and welfare of people who volunteer for research. it does this in two phases: reviewing the study before it begins and monitoring it periodically while it’s underway. Irb processes are integral to every stage of a clinical trial, from protocol development and initial submission to amendments and final reporting. mastery of irb requirements ensures that ethical principles are built into study design from the ground up. Formal review procedures for institutional human subject studies were originally developed in direct response to research abuses in the 20th century. Understand what an irb is, what it does, and how it works. learn about the federal and regulatory guidelines, timelines, and history of institutional review boards. The irb approval process the irb approval process is a structured procedure designed to ensure that all research involving human subjects meets stringent ethical and regulatory standards. researchers must submit a comprehensive application to the irb, detailing every aspect of their proposed study. The u m institutional review boards (irbs) fulfill their goals to protect human research participants and support the design and conduct of sound research by reviewing and approving irb submissions for new applications, amendments, and continuing reviews.
Comments are closed.