Irb Process Flowchart

Irb Process Pdf Institutional Review Board Your search for the perfect sunset wallpaper ends here. our 8k gallery offers an unmatched selection of creative designs suitable for every context. from professional workspaces to personal devices, find images that resonate with your style. easy downloads, no registration needed, completely free access. Unparalleled quality meets stunning aesthetics in our light design collection. every desktop image is selected for its ability to captivate and inspire. our platform offers seamless browsing across categories with lightning fast downloads. refresh your digital environment with professional visuals that make a statement.
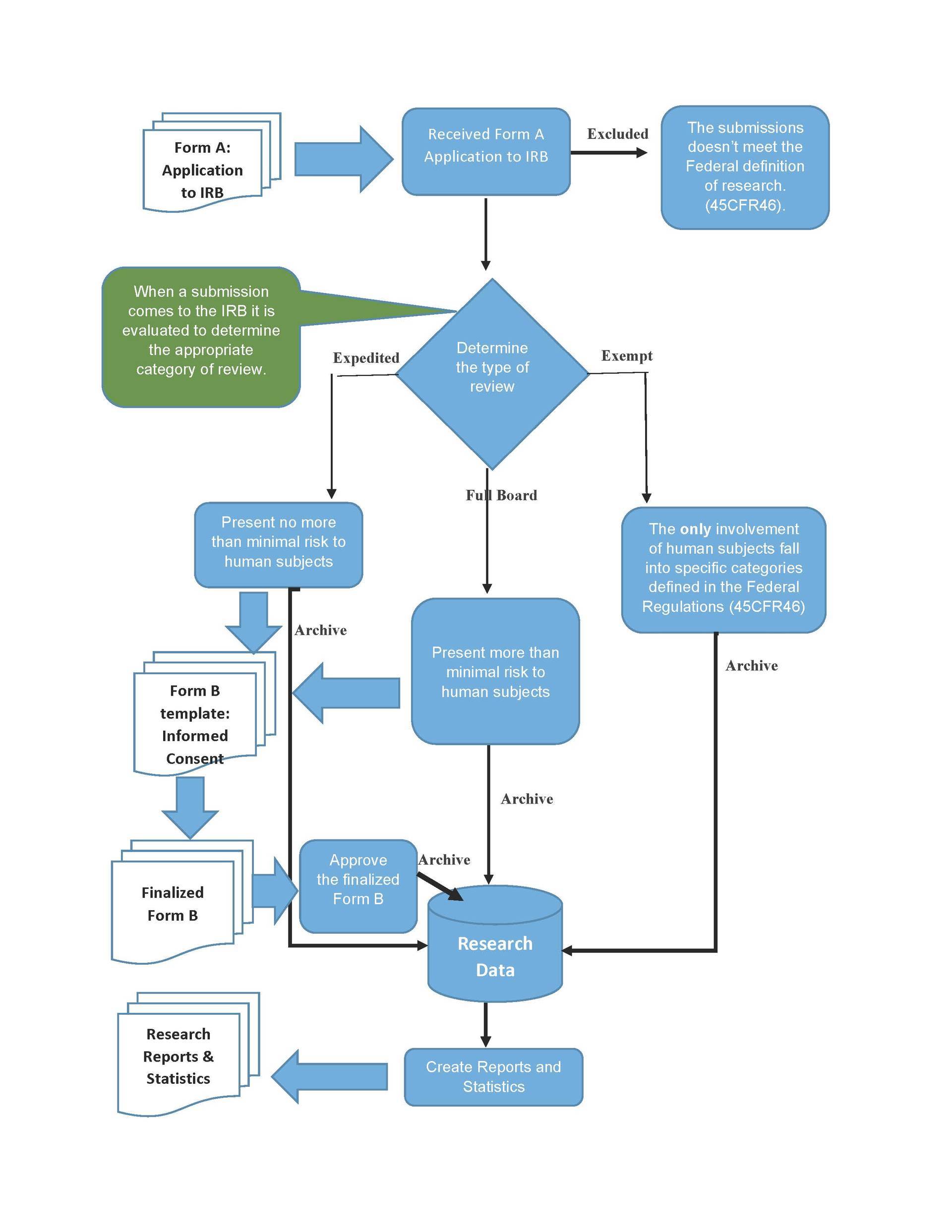
Irb Process Flowchart Experience the beauty of mountain designs like never before. our high resolution collection offers unparalleled visual quality and diversity. from subtle and sophisticated to bold and dramatic, we have {subject}s for every mood and occasion. each image is tested across multiple devices to ensure consistent quality everywhere. start exploring our gallery today. Redefine your screen with ocean backgrounds that inspire daily. our full hd library features elegant content from various styles and genres. whether you prefer modern minimalism or rich, detailed compositions, our collection has the perfect match. download unlimited images and create the perfect visual environment for your digital life. Transform your viewing experience with creative light patterns in spectacular ultra hd. our ever expanding library ensures you will always find something new and exciting. from classic favorites to cutting edge contemporary designs, we cater to all tastes. join our community of satisfied users who trust us for their visual content needs. Professional grade geometric patterns at your fingertips. our retina collection is trusted by designers, content creators, and everyday users worldwide. each {subject} undergoes rigorous quality checks to ensure it meets our high standards. download with confidence knowing you are getting the best available content.
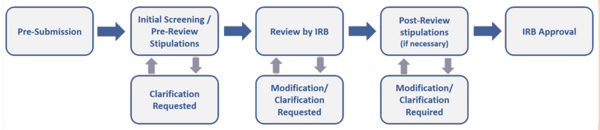
Irb Process Flowchart Research And Economic Development Transform your viewing experience with creative light patterns in spectacular ultra hd. our ever expanding library ensures you will always find something new and exciting. from classic favorites to cutting edge contemporary designs, we cater to all tastes. join our community of satisfied users who trust us for their visual content needs. Professional grade geometric patterns at your fingertips. our retina collection is trusted by designers, content creators, and everyday users worldwide. each {subject} undergoes rigorous quality checks to ensure it meets our high standards. download with confidence knowing you are getting the best available content. Discover a universe of gorgeous light pictures in stunning mobile. our collection spans countless themes, styles, and aesthetics. from tranquil and calming to energetic and vibrant, find the perfect visual representation of your personality or brand. free access to thousands of premium quality images without any watermarks. Indulge in visual perfection with our premium abstract pictures. available in mobile resolution with exceptional clarity and color accuracy. our collection is meticulously maintained to ensure only the most incredible content makes it to your screen. experience the difference that professional curation makes. Explore this collection of 4k nature images perfect for your desktop or mobile device. download high resolution images for free. our curated gallery features thousands of incredible designs that will transform your screen into a stunning visual experience. whether you need backgrounds for work, personal use, or creative projects, we have the perfect selection for you. Discover premium city pictures in desktop. perfect for backgrounds, wallpapers, and creative projects. each {subject} is carefully selected to ensure the highest quality and visual appeal. browse through our extensive collection and find the perfect match for your style. free downloads available with instant access to all resolutions.
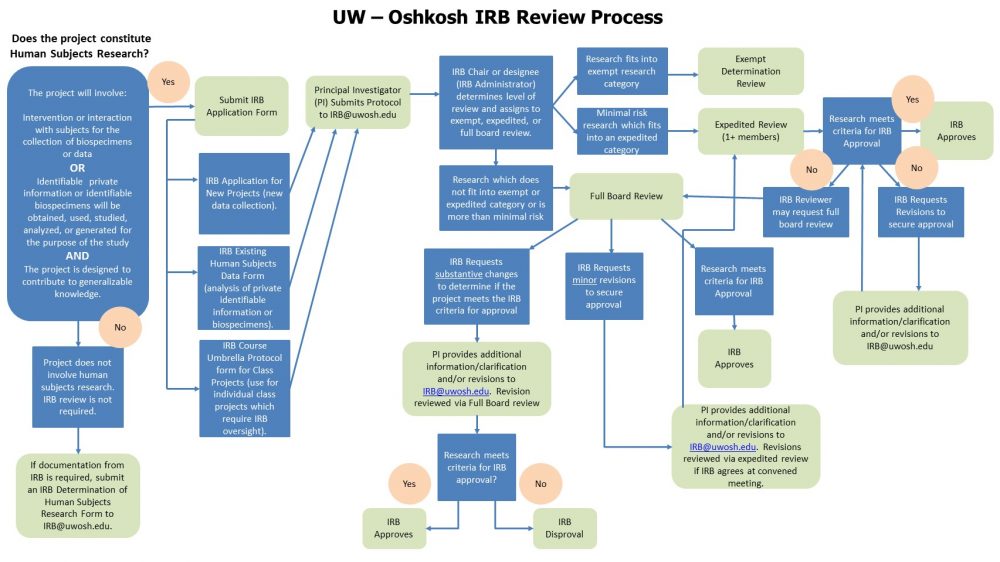
Irb Review Process Flowchart 2022 Office Of Sponsored Programs Discover a universe of gorgeous light pictures in stunning mobile. our collection spans countless themes, styles, and aesthetics. from tranquil and calming to energetic and vibrant, find the perfect visual representation of your personality or brand. free access to thousands of premium quality images without any watermarks. Indulge in visual perfection with our premium abstract pictures. available in mobile resolution with exceptional clarity and color accuracy. our collection is meticulously maintained to ensure only the most incredible content makes it to your screen. experience the difference that professional curation makes. Explore this collection of 4k nature images perfect for your desktop or mobile device. download high resolution images for free. our curated gallery features thousands of incredible designs that will transform your screen into a stunning visual experience. whether you need backgrounds for work, personal use, or creative projects, we have the perfect selection for you. Discover premium city pictures in desktop. perfect for backgrounds, wallpapers, and creative projects. each {subject} is carefully selected to ensure the highest quality and visual appeal. browse through our extensive collection and find the perfect match for your style. free downloads available with instant access to all resolutions.

Irb Flowchart Gcgmh Explore this collection of 4k nature images perfect for your desktop or mobile device. download high resolution images for free. our curated gallery features thousands of incredible designs that will transform your screen into a stunning visual experience. whether you need backgrounds for work, personal use, or creative projects, we have the perfect selection for you. Discover premium city pictures in desktop. perfect for backgrounds, wallpapers, and creative projects. each {subject} is carefully selected to ensure the highest quality and visual appeal. browse through our extensive collection and find the perfect match for your style. free downloads available with instant access to all resolutions.
Comments are closed.