Institutional Review Board Irb Psu
Institutional Review Board Irb Psu The human research protection program (hrpp) is the irb office for the entire university and provides support for all penn state researchers, including those at the college of medicine and penn state health. การประชุมคณะกรรมการจริยธรรมการวิจัยในมนุษย์ สาขาสังคม.
Institutional Review Board Irb Psu This session offers an overview of the prince sultan university institutional review board (psu irb) including irb process and any questions. this session also discusses some of the important checklists you need to follow to get it done quickly. Click “my inbox” on the left of the workspace. click “create new study” to display the study submission form. complete all questions applicable to your research; questions with a red asterisk are required. All items of the psu irb application form must be completed, and the form must be typed and submitted electronically (with signature) to the psu institutional review board ([email protected]). What's an irb? an institutional review board (irb) is a federally mandated entity that oversees the protection of human subjects in research. penn state has two irbs: the general review board and the noncompliance board.
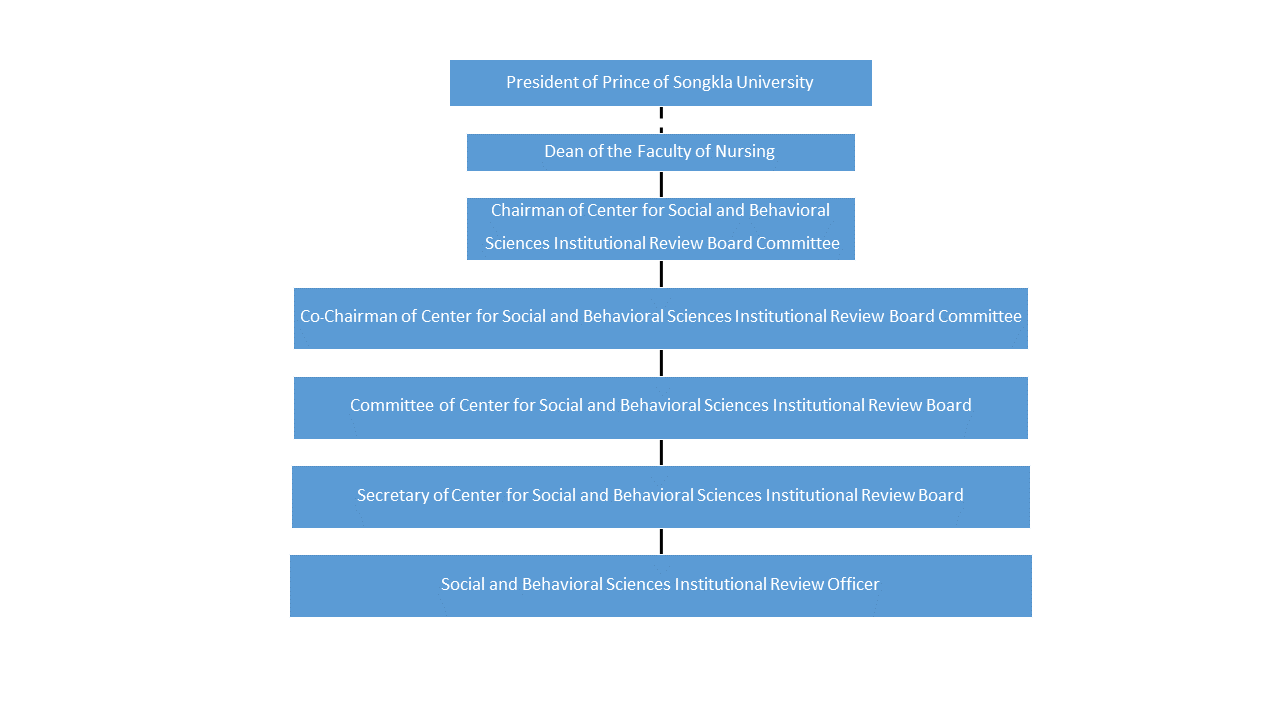
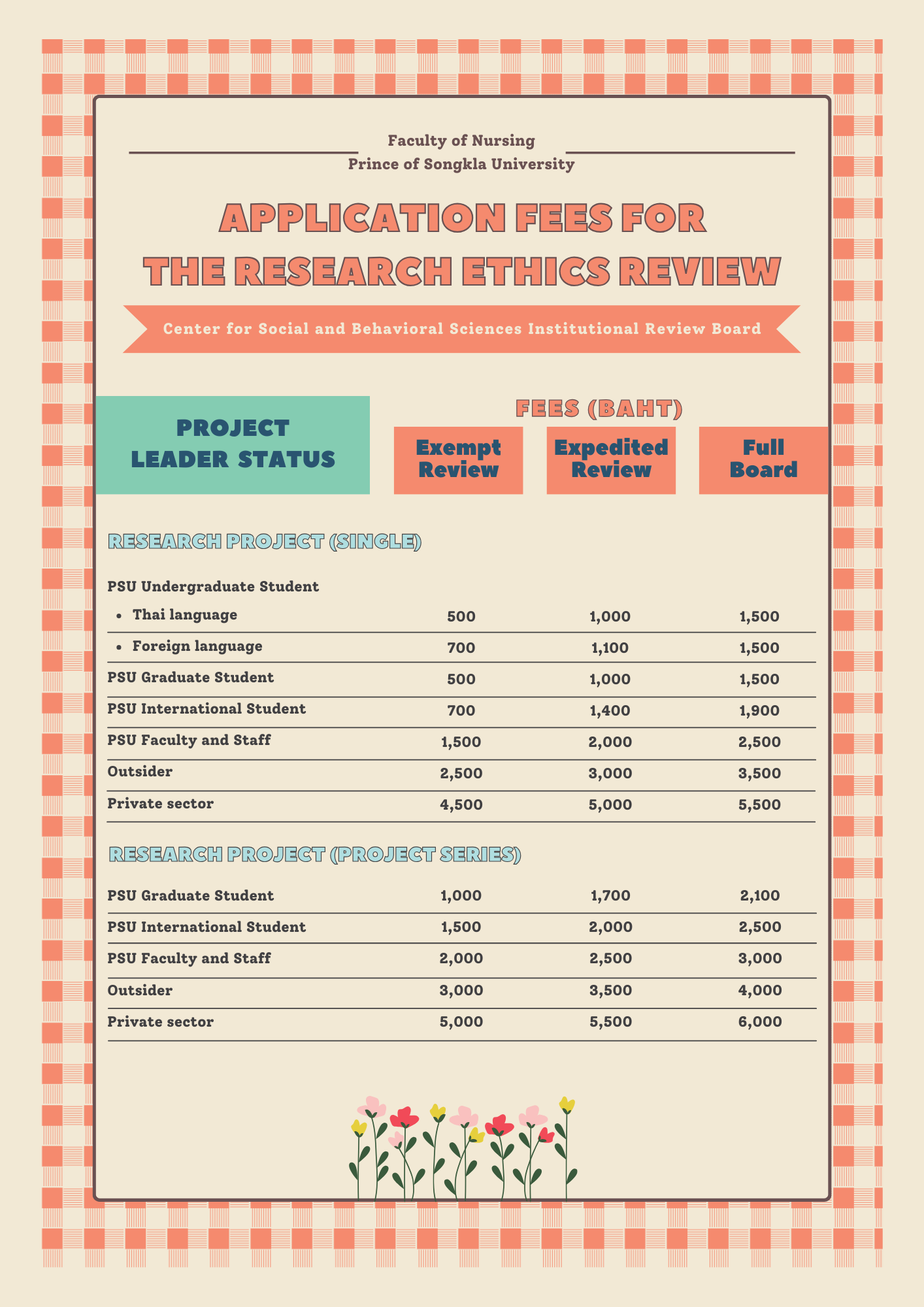
Institutional Review Board Irb Psu All items of the psu irb application form must be completed, and the form must be typed and submitted electronically (with signature) to the psu institutional review board ([email protected]). What's an irb? an institutional review board (irb) is a federally mandated entity that oversees the protection of human subjects in research. penn state has two irbs: the general review board and the noncompliance board. Attention: the center of social and behavioral sciences institutional review board committee, prince of songkla university has periodically updated the form to be consistent with its operations. The office for research protections (orp) ensures that research at the university is conducted in accordance with federal, state, and local regulations and guidelines that protect human participants, animals, students, and personnel involved with research. Sbsirb psu home about us information for researcher download regularity directions publish contact us th. All penn state employees and students conducting activities that meet the definition of both “research” and “human subject” must submit for institutional review board (irb) approval before beginning any research activity. irb approval cannot be retroactive.

Prince Sultan University Psu Institutional Review Board Attention: the center of social and behavioral sciences institutional review board committee, prince of songkla university has periodically updated the form to be consistent with its operations. The office for research protections (orp) ensures that research at the university is conducted in accordance with federal, state, and local regulations and guidelines that protect human participants, animals, students, and personnel involved with research. Sbsirb psu home about us information for researcher download regularity directions publish contact us th. All penn state employees and students conducting activities that meet the definition of both “research” and “human subject” must submit for institutional review board (irb) approval before beginning any research activity. irb approval cannot be retroactive.

Prince Sultan University Psu Instituational Review Board Irb Sbsirb psu home about us information for researcher download regularity directions publish contact us th. All penn state employees and students conducting activities that meet the definition of both “research” and “human subject” must submit for institutional review board (irb) approval before beginning any research activity. irb approval cannot be retroactive.

Institutional Review Board Irb Explained Ccrps
Comments are closed.