Irb Organizational Chart Gcgmh

Irb Organizational Chart Gcgmh We use cookies to ensure that we give you the best experience on our website. by continuing to use our site, we presume you have read and accepted our privacy notice. Gcgmh institutional review board is now at our official website (gcgmh.gov.ph) bringing ethics review accessible to researchers. gcgmh irb is the only phreb level ii accredited research ethics committee in the province of bohol.

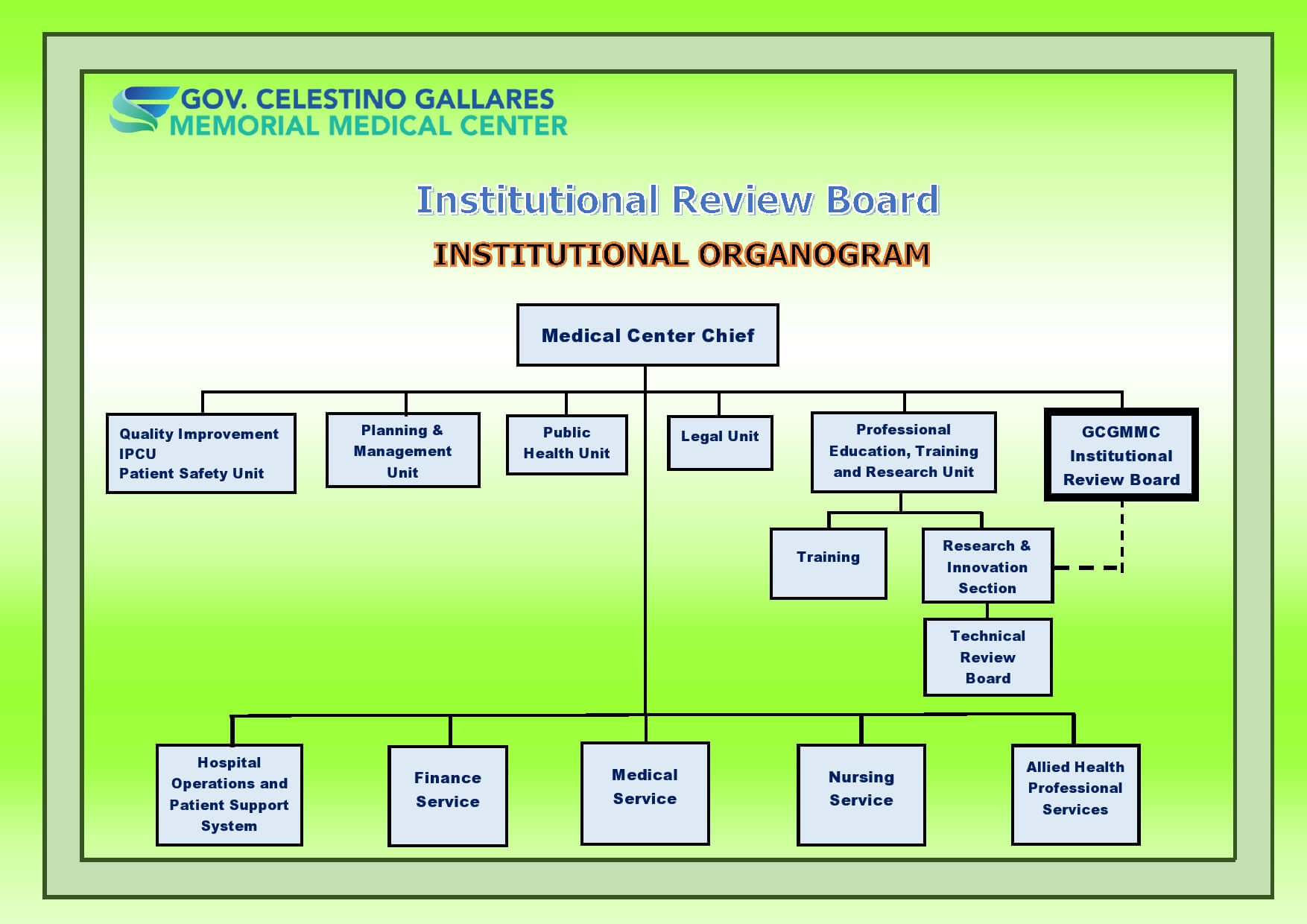
Irb Organizational Chart Gcgmh Position on gcgmh irb: secretariat. Membership to the irb was increased from 7 to 15 to include doctors representing the four major clinical areas (internal medicine, ob gyne, pedia, surgery and family medicine), 2 nurses and 3 non scientific and 2 non affiliated members. Irb org chart patient's corner bid opportunities career opportunities fees and rates galleries ipcu gad corner irb praise committee citizen's charter manual you are here: home irb institutional review board. Irbs are operated by iorgs, and each iorg can register one or more irbs. an initial registration registers both the iorg and its irb or irbs. updates are renewals, and change information for: the iorg, an already registered irb, or, registering additional irb (s) .
Irb Organizational Chart Gcgmh Irb org chart patient's corner bid opportunities career opportunities fees and rates galleries ipcu gad corner irb praise committee citizen's charter manual you are here: home irb institutional review board. Irbs are operated by iorgs, and each iorg can register one or more irbs. an initial registration registers both the iorg and its irb or irbs. updates are renewals, and change information for: the iorg, an already registered irb, or, registering additional irb (s) . Irb organizational chart oleg kisselev, phd chair, boards #1 & #2 administrative chair. In july 2017, an sop workshop was conducted for the members of the mmerc. review of health related research and surveying and evaluating ethical review practices, ec irb standard operating procedures (sops), local regulations, and standards in ethical review. The database of registered iorgs and irbs includes information on irbs that are regulated by ohrp only, ohrp and fda , and fda only. 長庚獨立審查案件,一般審查不需送衛福部 (a3)案件:執行院區為林口單中心案件的新案、期中、變更案、結案、研究不遵從事件、嚴重不良事件暨非預期問題、暫停、備查、終止、撤案、實地稽核審理作業。 2. 長庚獨立審查案件,一般審查不需送衛福部 (a3)、簡易審查 (b0 b1)類案件:執行院區為土城、長庚診所、台北、桃園 (含桃園護理之家)、基隆、基隆情人湖單中心案件的新案、期中、變更案、結案、研究不遵從事件、嚴重不良事件暨非預期問題、暫停、備查、終止、撤案、實地稽核審理作業。 3. irb教育訓練統籌及規劃、長庚學習網irb線上教育課程。 4. his臨床執行管控系統維護。 5. clinicaltrials.gov登錄與管理。.
Comments are closed.