Atomic Orbitals Explained Domainloki
Atomic Orbitals Explained Iigerty The main difference between atomic and molecular orbital is that the electrons in an atomic orbital are influenced by one positive nucleus, while the electrons of a molecular orbital are influenced by the two or more nuclei depending upon the number of atoms in a molecule. Explore atomic orbitals—s, p, d, and f shapes—and how they describe electron behavior in the modern atomic model. includes visuals and key characteristics.

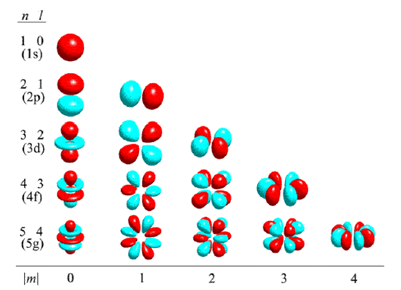
Atomic Orbitals Explained Fun Learning Explained Chemistry Learn about atomic orbitals with diagrams of their types, shapes, energy, and a chart of their filling orders. Atomic orbitals are basic building blocks of the atomic orbital model (or electron cloud or wave mechanics model), a modern framework for visualizing submicroscopic behavior of electrons in matter. An atom is a system of one or more electrons bound to a nucleus. here is a quantum microscope image of a hydrogen atom:. This section explains atomic orbitals, emphasizing their quantum mechanical nature compared to bohr's orbits. it covers the order and energy levels of orbitals from 1s to 3d and details s and p ….
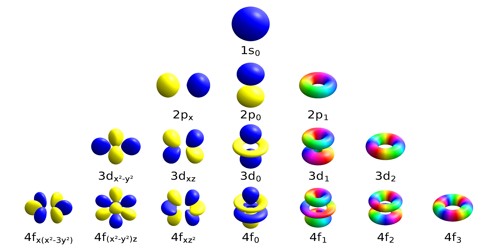
Atomic Orbitals Explained Domainloki An atom is a system of one or more electrons bound to a nucleus. here is a quantum microscope image of a hydrogen atom:. This section explains atomic orbitals, emphasizing their quantum mechanical nature compared to bohr's orbits. it covers the order and energy levels of orbitals from 1s to 3d and details s and p …. Master atomic orbitals with clear explanations, diagrams, and real examples. start learning now with vedantu. An atomic orbital is a region of space around the nucleus where the probability of finding an electron is highest. instead of moving in fixed circular paths around the nucleus, electrons exist in these regions according to the principles of quantum mechanics. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). within each shell of an atom there are some combinations of orbitals. While electron shells and orbitals are closely related, orbitals provide a more accurate picture of the electron configuration of an atom. that’s because orbitals actually specify the shape and position of the regions of space that electrons occupy.

Atomic Orbitals Explained Vectorsand Master atomic orbitals with clear explanations, diagrams, and real examples. start learning now with vedantu. An atomic orbital is a region of space around the nucleus where the probability of finding an electron is highest. instead of moving in fixed circular paths around the nucleus, electrons exist in these regions according to the principles of quantum mechanics. There are four types of orbitals that you should be familiar with s, p, d and f (sharp, principle, diffuse and fundamental). within each shell of an atom there are some combinations of orbitals. While electron shells and orbitals are closely related, orbitals provide a more accurate picture of the electron configuration of an atom. that’s because orbitals actually specify the shape and position of the regions of space that electrons occupy.
Comments are closed.