Atomic Orbitals Explained Fun Learning Explained Chemistry

Atomic Orbitals Explained Shapes Energy Rules Explore atomic orbitals—s, p, d, and f shapes—and how they describe electron behavior in the modern atomic model. includes visuals and key characteristics. This section explains atomic structure and the role of orbitals in electron configuration. it describes the different types of atomic orbitals (s, p, d, f) and their shapes, energies, and how they ….
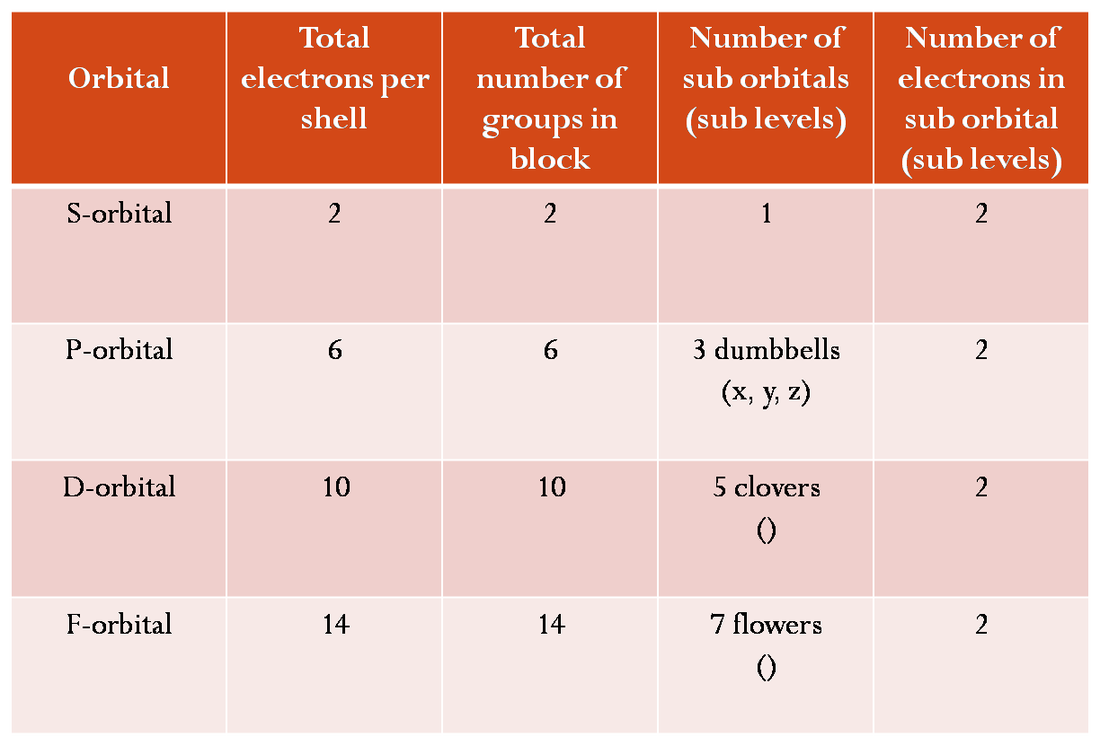
Atomic Orbitals And Aufbau Ssc Chemistry Electronjump is a free online educational puzzle platformer game designed for high school and college chemistry students to learn orbital diagrams, electron configurations, quantum numbers, and more through engaging gameplay. Master atomic orbitals with clear explanations, diagrams, and real examples. start learning now with vedantu. Atomic orbitals are regions around the nucleus where electrons are most likely to be found. these orbitals differ in shape, size, energy, and the number of electrons they can hold. Atomic orbitals are basic building blocks of the atomic orbital model (or electron cloud or wave mechanics model), a modern framework for visualizing submicroscopic behavior of electrons in matter.
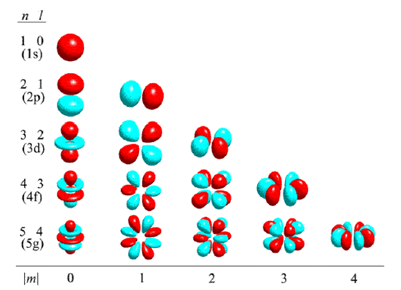
Atomic Orbitals Explained Iigerty Atomic orbitals are regions around the nucleus where electrons are most likely to be found. these orbitals differ in shape, size, energy, and the number of electrons they can hold. Atomic orbitals are basic building blocks of the atomic orbital model (or electron cloud or wave mechanics model), a modern framework for visualizing submicroscopic behavior of electrons in matter. What are atomic orbitals, and why do they matter in chemistry? this short, visual video breaks down the concept of atomic orbitals—from s, p, d, and f shapes to how they relate to. According to atomic theory and quantum mechanics, an atomic orbital is a mathematical expression that presents the wave like behaviour of 1 or 2 electrons of an atom. unlike the fixed orbit path of planets around the sun, electrons present in an atom don't follow a definite path. Learn about atomic orbitals with diagrams of their types, shapes, energy, and a chart of their filling orders. There are four different kinds of orbitals, denoted s, p, d, and f, each with a different shape. of the four, we’ll be concerned primarily with s and p orbitals because these are the most common in organic and biological chemistry.
Comments are closed.