Atomic Orbitals Explained Vectorsand

Atomic Orbitals Explained Bezyself Explore atomic orbitals—s, p, d, and f shapes—and how they describe electron behavior in the modern atomic model. includes visuals and key characteristics. Each orbital in an atom is characterized by a set of values of three quantum numbers n, ℓ, and mℓ, which respectively correspond to an electron's energy, its orbital angular momentum, and its orbital angular momentum projected along a chosen axis (magnetic quantum number).

Atomic Orbitals Explained Sendmilo This section explains atomic orbitals, emphasizing their quantum mechanical nature compared to bohr's orbits. it covers the order and energy levels of orbitals from 1s to 3d and details s and p …. Learn about atomic orbitals with diagrams of their types, shapes, energy, and a chart of their filling orders. That probability cloud is called an orbital (not orbit): orbital: a region where there is a 90% chance of finding an electron. the simplest orbital is the "s" orbital (such as the single electron in hydrogen): but we can have more than one orbital, each with a different energy state. According to atomic theory and quantum mechanics, an atomic orbital is a mathematical expression that presents the wave like behaviour of 1 or 2 electrons of an atom. unlike the fixed orbit path of planets around the sun, electrons present in an atom don't follow a definite path.
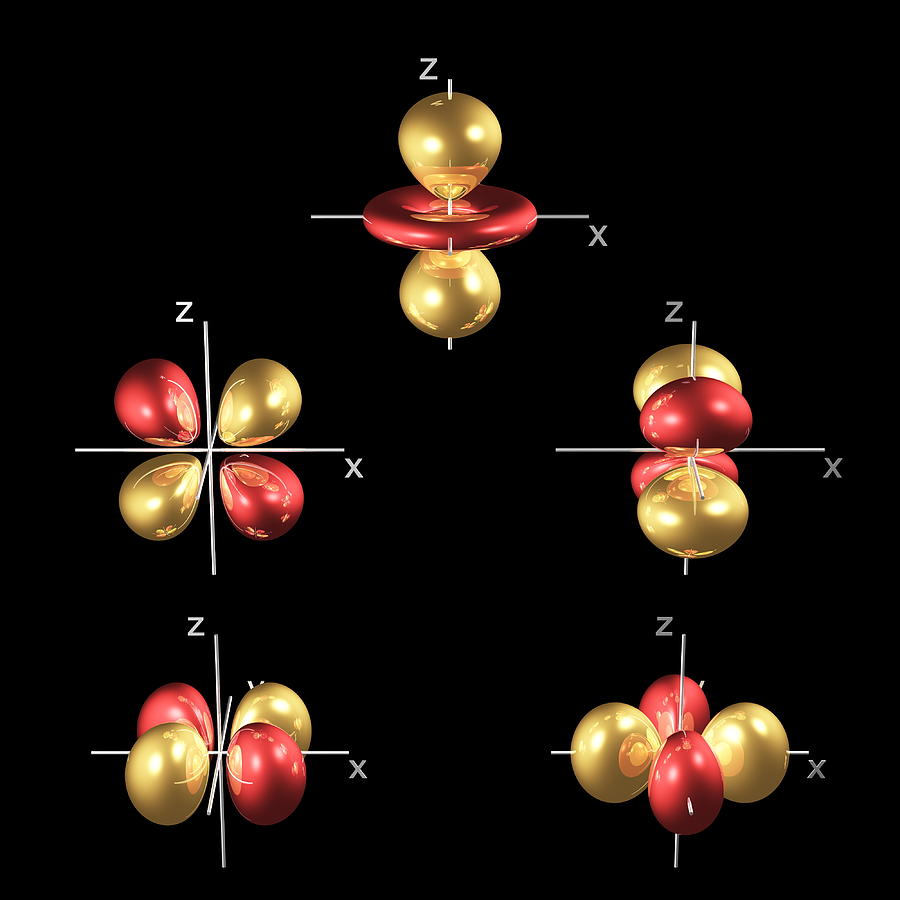
Atomic Orbitals Explained Garagebro That probability cloud is called an orbital (not orbit): orbital: a region where there is a 90% chance of finding an electron. the simplest orbital is the "s" orbital (such as the single electron in hydrogen): but we can have more than one orbital, each with a different energy state. According to atomic theory and quantum mechanics, an atomic orbital is a mathematical expression that presents the wave like behaviour of 1 or 2 electrons of an atom. unlike the fixed orbit path of planets around the sun, electrons present in an atom don't follow a definite path. Atomic orbitals are regions around the nucleus where electrons are most likely to be found. these orbitals differ in shape, size, energy, and the number of electrons they can hold. Master atomic orbitals with clear explanations, diagrams, and real examples. start learning now with vedantu. Understand atomic orbitals: what they are, why they have shapes, how electrons fill them, and how this predicts configuration, bonding, and trends. Atomic orbitals are the basic building blocks of the atomic orbital model (alternatively known as the electron cloud or wave mechanics model), a modern framework for visualizing the microscopic behavior of electrons in matter.

Atomic Orbitals Explained Vectorsand Atomic orbitals are regions around the nucleus where electrons are most likely to be found. these orbitals differ in shape, size, energy, and the number of electrons they can hold. Master atomic orbitals with clear explanations, diagrams, and real examples. start learning now with vedantu. Understand atomic orbitals: what they are, why they have shapes, how electrons fill them, and how this predicts configuration, bonding, and trends. Atomic orbitals are the basic building blocks of the atomic orbital model (alternatively known as the electron cloud or wave mechanics model), a modern framework for visualizing the microscopic behavior of electrons in matter.
Comments are closed.