Orbitals Atomic Energy Levels Sublevels Explained Shell Electron

Main Energy Levels Sublevels And Orbitals Pdf Atomic Orbital Each energy level (shell) contains sub shells: s, p, d (and f from n = 4). each sub shell consists of orbitals, which are regions of space where there is a high probability of finding an electron. Energy levels, sublevels, orbitals, and suborbitals define the structured way electrons arrange within an atom. each term describes a specific aspect of how electrons occupy space and energy.
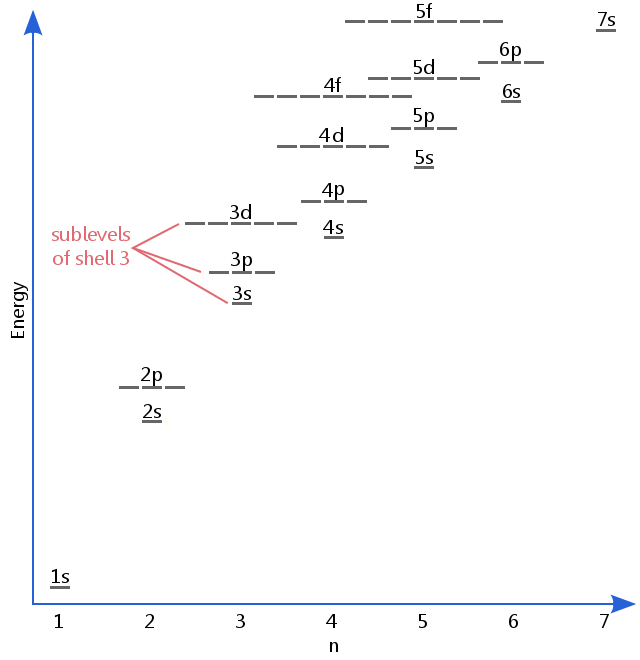
Orbitals Atomic Energy Levels Sublevels Explained Shell Electron In this lesson you will learn that electrons reside in principal energy levels (n = 1, 2, 3…) and sublevels (s, p, d, f), where each orbital type has a distinct energy. The electrons in an atom revolve around the nucleus along fixed circular routes, which are known as the energy levels, orbits, or shells of the atom. in their orbits, electrons don’t gain or lose energy. Electrons are attracted to the positively charged nucleus of an atom and exist in specific regions associated with defined energy levels. these regions are organized into shells, sub shells, and orbitals, each describing the energy, position, and probability of finding an electron. Electrons fill principal energy levels, or shells, to have the lowest possible energy. within the shells, they also fill the sublevels s, p, d, and f to have the lowest energy. so, electrons in orbitals in the s sublevel, called s orbitals, have less energy than those in p sublevel ….

Orbitals Atomic Energy Levels Sublevels Explained Shell Electron Electrons are attracted to the positively charged nucleus of an atom and exist in specific regions associated with defined energy levels. these regions are organized into shells, sub shells, and orbitals, each describing the energy, position, and probability of finding an electron. Electrons fill principal energy levels, or shells, to have the lowest possible energy. within the shells, they also fill the sublevels s, p, d, and f to have the lowest energy. so, electrons in orbitals in the s sublevel, called s orbitals, have less energy than those in p sublevel …. This page explains electron organization within atoms based on quantum mechanics, detailing shells and subshells defined by energy levels and quantum numbers. it covers electron configurations for …. An electron shell represents the primary energy level of an atom, designated by the principal quantum number (n). within each shell, there are sublevels (labeled s, p, d, and f), which are specific divisions that define the shape and spatial distribution of the orbitals. Learn about electron shells and orbitals for your a level chemistry exam. find information on quantum numbers, subshells and orbital shapes. Within each main energy level are sublevels, represented by letters, that further define the possible energies of electrons. orbitals, which can each hold up to 2 electrons, describe the specific locations where electrons are probably arranged in space within an atom.
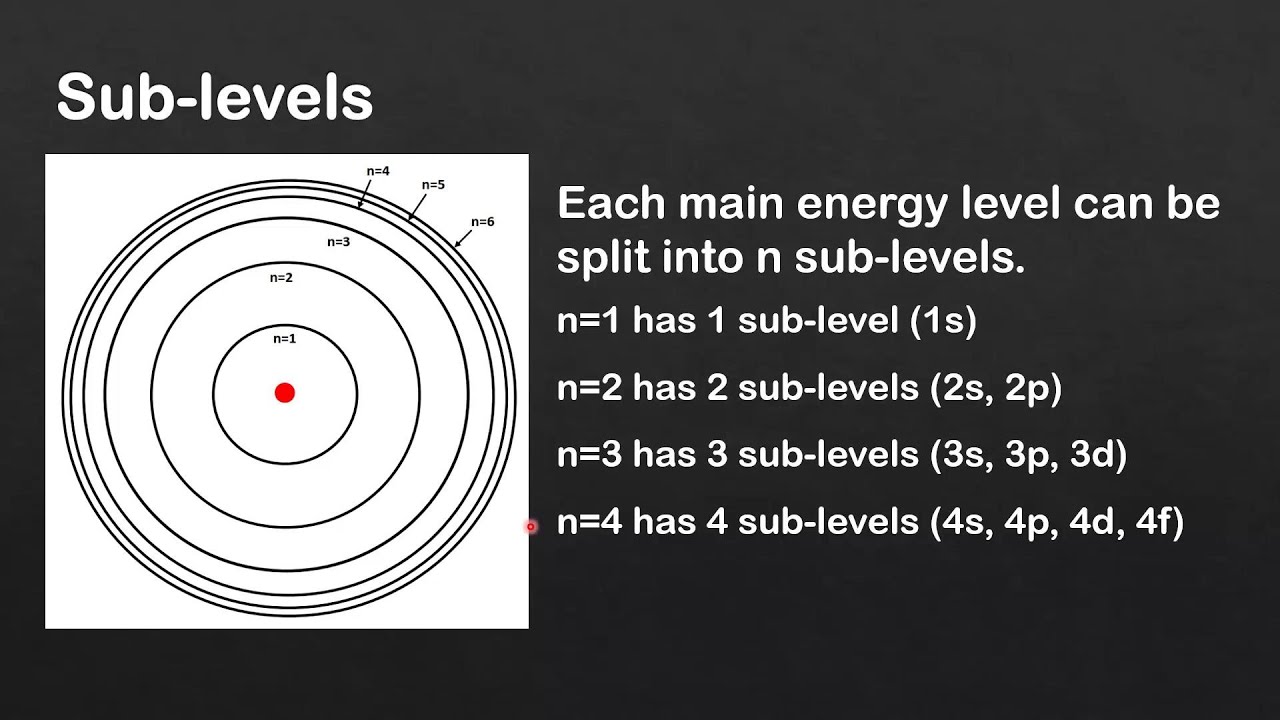
Orbitals Atomic Energy Levels Sublevels Explained Shell Electron This page explains electron organization within atoms based on quantum mechanics, detailing shells and subshells defined by energy levels and quantum numbers. it covers electron configurations for …. An electron shell represents the primary energy level of an atom, designated by the principal quantum number (n). within each shell, there are sublevels (labeled s, p, d, and f), which are specific divisions that define the shape and spatial distribution of the orbitals. Learn about electron shells and orbitals for your a level chemistry exam. find information on quantum numbers, subshells and orbital shapes. Within each main energy level are sublevels, represented by letters, that further define the possible energies of electrons. orbitals, which can each hold up to 2 electrons, describe the specific locations where electrons are probably arranged in space within an atom.
Comments are closed.