Atomic Orbitals Explained Garagebro
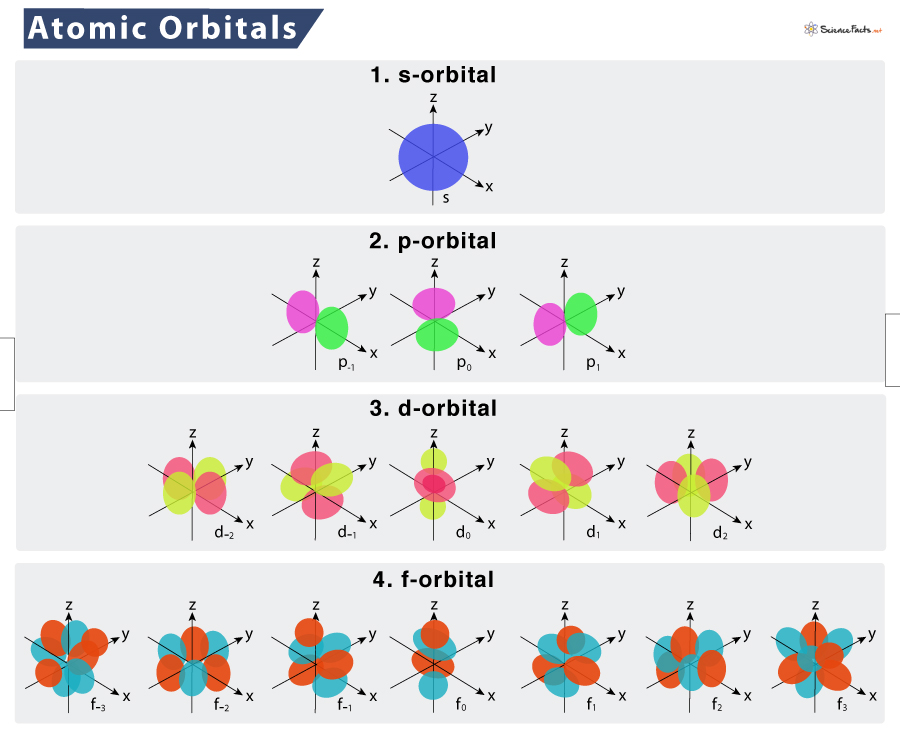
Atomic Orbital Definition Types Shapes And Diagram Students will first look at a diagram and animation to understand the basic pattern of the arrangement of electrons on energy levels around an atom. students will again focus on the first 20 elements. Explore atomic orbitals—s, p, d, and f shapes—and how they describe electron behavior in the modern atomic model. includes visuals and key characteristics.

Overview Of Atomic Orbitals Stock Photo 340098275 Shutterstock That probability cloud is called an orbital (not orbit): orbital: a region where there is a 90% chance of finding an electron. the simplest orbital is the "s" orbital (such as the single electron in hydrogen): but we can have more than one orbital, each with a different energy state. This section explains atomic orbitals, emphasizing their quantum mechanical nature compared to bohr's orbits. it covers the order and energy levels of orbitals from 1s to 3d and details s and p …. While electron shells and orbitals are closely related, orbitals provide a more accurate picture of the electron configuration of an atom. that’s because orbitals actually specify the shape and position of the regions of space that electrons occupy. Learn about atomic orbitals with diagrams of their types, shapes, energy, and a chart of their filling orders.

Shapes Of Atomic Orbitals While electron shells and orbitals are closely related, orbitals provide a more accurate picture of the electron configuration of an atom. that’s because orbitals actually specify the shape and position of the regions of space that electrons occupy. Learn about atomic orbitals with diagrams of their types, shapes, energy, and a chart of their filling orders. According to atomic theory and quantum mechanics, an atomic orbital is a mathematical expression that presents the wave like behaviour of 1 or 2 electrons of an atom. unlike the fixed orbit path of planets around the sun, electrons present in an atom don't follow a definite path. What is an atomic orbital in chemistry? an atomic orbital is a mathematical function, known as a wave function, that describes the three dimensional region around an atom's nucleus where there is the highest probability of finding an electron. In this lesson, we will learn. atomic orbitals are regions of space in which electrons can be found. each orbital can fit two electrons and different orbitals have different shapes. the s sub level has one spherically shaped orbital, while the p sub level has three dumbbell shaped orbitals. Understand atomic orbitals: what they are, why they have shapes, how electrons fill them, and how this predicts configuration, bonding, and trends.

Atomic Orbitals Explained S P D And F Shapes According to atomic theory and quantum mechanics, an atomic orbital is a mathematical expression that presents the wave like behaviour of 1 or 2 electrons of an atom. unlike the fixed orbit path of planets around the sun, electrons present in an atom don't follow a definite path. What is an atomic orbital in chemistry? an atomic orbital is a mathematical function, known as a wave function, that describes the three dimensional region around an atom's nucleus where there is the highest probability of finding an electron. In this lesson, we will learn. atomic orbitals are regions of space in which electrons can be found. each orbital can fit two electrons and different orbitals have different shapes. the s sub level has one spherically shaped orbital, while the p sub level has three dumbbell shaped orbitals. Understand atomic orbitals: what they are, why they have shapes, how electrons fill them, and how this predicts configuration, bonding, and trends.
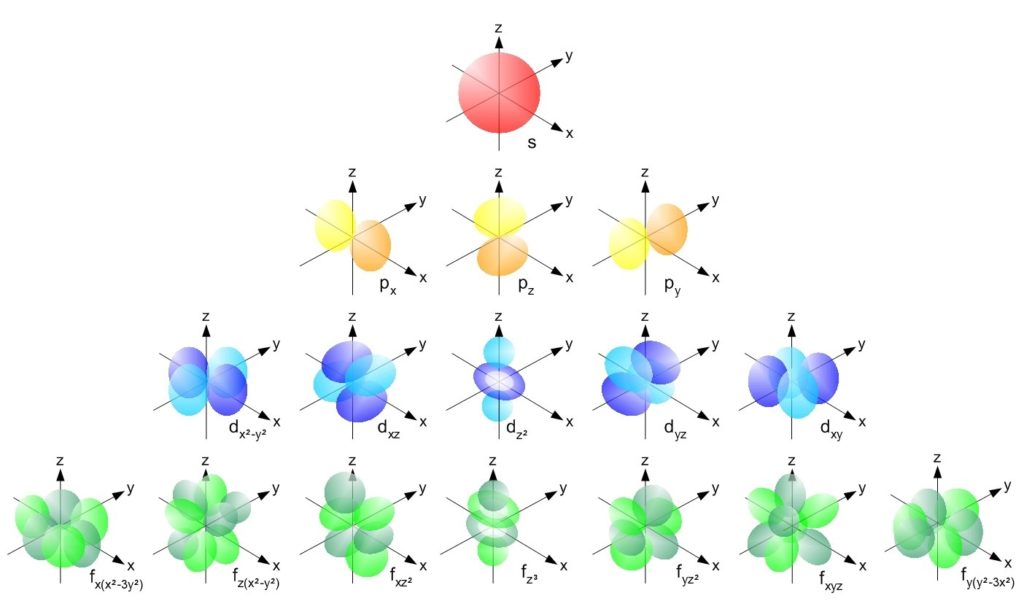
Atomic Orbital Types Energies Diagram Examples Summary In this lesson, we will learn. atomic orbitals are regions of space in which electrons can be found. each orbital can fit two electrons and different orbitals have different shapes. the s sub level has one spherically shaped orbital, while the p sub level has three dumbbell shaped orbitals. Understand atomic orbitals: what they are, why they have shapes, how electrons fill them, and how this predicts configuration, bonding, and trends.

Shapes Of Atomic Orbitals Overview Examples Expii
Comments are closed.