Atomic Orbitals Explained Shapes Energy Rules
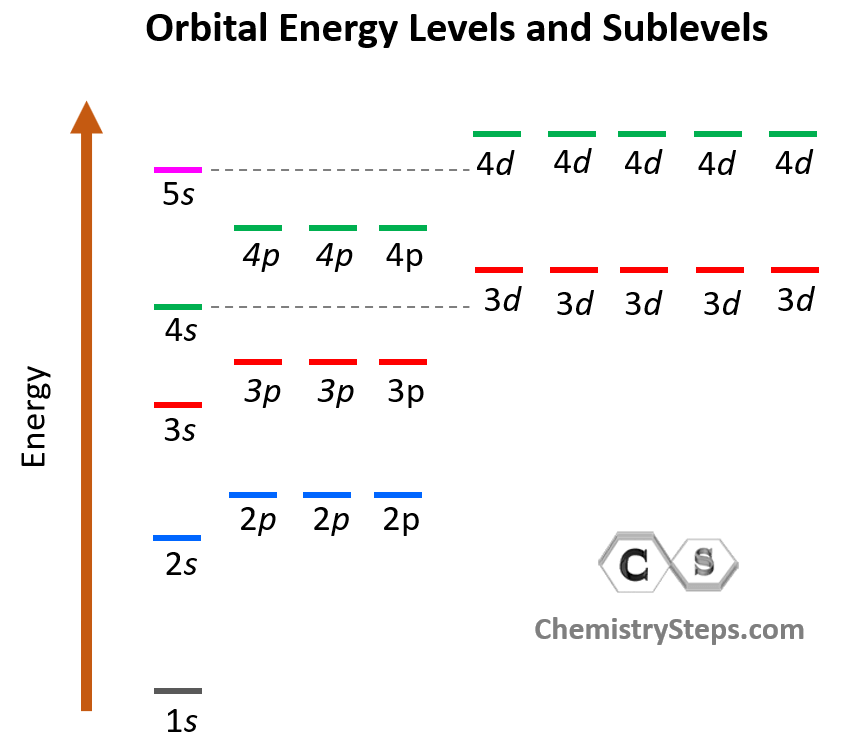
Orbitals Atomic Energy Levels Sublevels Explained Shell Electron Explore atomic orbitals—s, p, d, and f shapes—and how they describe electron behavior in the modern atomic model. includes visuals and key characteristics. Electronic orbitals are regions within the atom in which electrons have the highest probability of being found. there are multiple orbitals within an atom. each has its own specific energy level and properties.

Shapes Of Atomic Orbitals Understand atomic orbitals: what they are, why they have shapes, how electrons fill them, and how this predicts configuration, bonding, and trends. Each orbital in an atom is characterized by a set of values of three quantum numbers n, ℓ, and mℓ, which respectively correspond to an electron's energy, its orbital angular momentum, and its orbital angular momentum projected along a chosen axis (magnetic quantum number). Learn about atomic orbitals with diagrams of their types, shapes, energy, and a chart of their filling orders. The orbitals in an atom are organized into different layers around the nucleus called electron shells, which are centered around the nucleus and have successively larger size and energy.

Figure 75 Atomic Orbitals Explained And Derived By Energy Learn about atomic orbitals with diagrams of their types, shapes, energy, and a chart of their filling orders. The orbitals in an atom are organized into different layers around the nucleus called electron shells, which are centered around the nucleus and have successively larger size and energy. Different orbitals have different shapes and energies. orbital types are labelled with letters – s, p, d and f. shells describe how far away an electron is from a nucleus, and therefore its energy. due to different shapes, orbitals within a shell can have different energies and form sub shells. Based on their shapes and energy levels, atomic orbitals are mainly classified into four types. these orbitals differ in their shapes, orientations, and the number of electrons they can hold. The energy of an orbital depends on both its size and its shape because the electron spends more of its time further from the nucleus of the atom as the orbital becomes larger or the shape becomes more complex. Energy levels, sublevels, orbitals, and suborbitals define the structured way electrons arrange within an atom. each term describes a specific aspect of how electrons occupy space and energy.
Figure 88 Atomic Orbitals Explained And Derived By Energy Different orbitals have different shapes and energies. orbital types are labelled with letters – s, p, d and f. shells describe how far away an electron is from a nucleus, and therefore its energy. due to different shapes, orbitals within a shell can have different energies and form sub shells. Based on their shapes and energy levels, atomic orbitals are mainly classified into four types. these orbitals differ in their shapes, orientations, and the number of electrons they can hold. The energy of an orbital depends on both its size and its shape because the electron spends more of its time further from the nucleus of the atom as the orbital becomes larger or the shape becomes more complex. Energy levels, sublevels, orbitals, and suborbitals define the structured way electrons arrange within an atom. each term describes a specific aspect of how electrons occupy space and energy.
Comments are closed.