Electronegativity Vs Electron Affinity

Electronegativity Vs Electron Affinity What S The Difference The best differentiating statement is: electronegativity describes an atom’s tendency to attract electrons within a chemical bond, while electron affinity is the energy released when a neutral atom gains an extra electron. Electronegativity and electron affinity are two separate chemical characteristics associated with elements. the major distinction between electronegativity and electron affinity is that electronegativity is the attribute associated to an electron’s capacity to attract towards an atom.

Electronegativity Vs Electron Affinity What S The Difference Unlike electron affinity, electronegativity is not a measure of energy released or absorbed. it is instead a relative measure of an atom’s electron pulling power within a molecular structure. Learn the difference between electron affinity and electronegativity, two properties that describe the attraction of electrons. electron affinity measures the energy change when an atom gains an electron, while electronegativity measures the attraction of electrons in a bond. Electronegativity is a qualitative measure of an atom’s ability to attract electrons within a chemical bond, while electron affinity is a quantitative measure of the energy change when an isolated atom gains an electron. Learn the difference between electronegativity and electron affinity, two chemical properties related to atom's ability to attract electrons. find definitions, examples, comparison table and quiz on this topic.

Electronegativity Vs Electron Affinity What S The Difference Electronegativity is a qualitative measure of an atom’s ability to attract electrons within a chemical bond, while electron affinity is a quantitative measure of the energy change when an isolated atom gains an electron. Learn the difference between electronegativity and electron affinity, two chemical properties related to atom's ability to attract electrons. find definitions, examples, comparison table and quiz on this topic. Electronegativity deals with individual atoms, while electron affinity deals with atoms in a molecule. electronegativity is also a property, while electron affinity is a measurement. Electronegativity is a chemical property that describes the ability of an atom to attract electrons towards itself in a chemical bond. while, electron affinity refers to the amount of energy released or absorbed when an electron is added to a neutral atom to form a negative ion. Electronegativity refers to the ability of an atom to attract shared electrons in a chemical bond, while electron affinity measures an atom’s tendency to accept an additional electron to form a negative ion. Electron affinity – a neutral atom can accept an electron to form a negative ion. in this process, in general, energy is released. – the electron affinity (ea) of an element is the amount of energy released when an electron is added to a gaseous atom to form an anion.
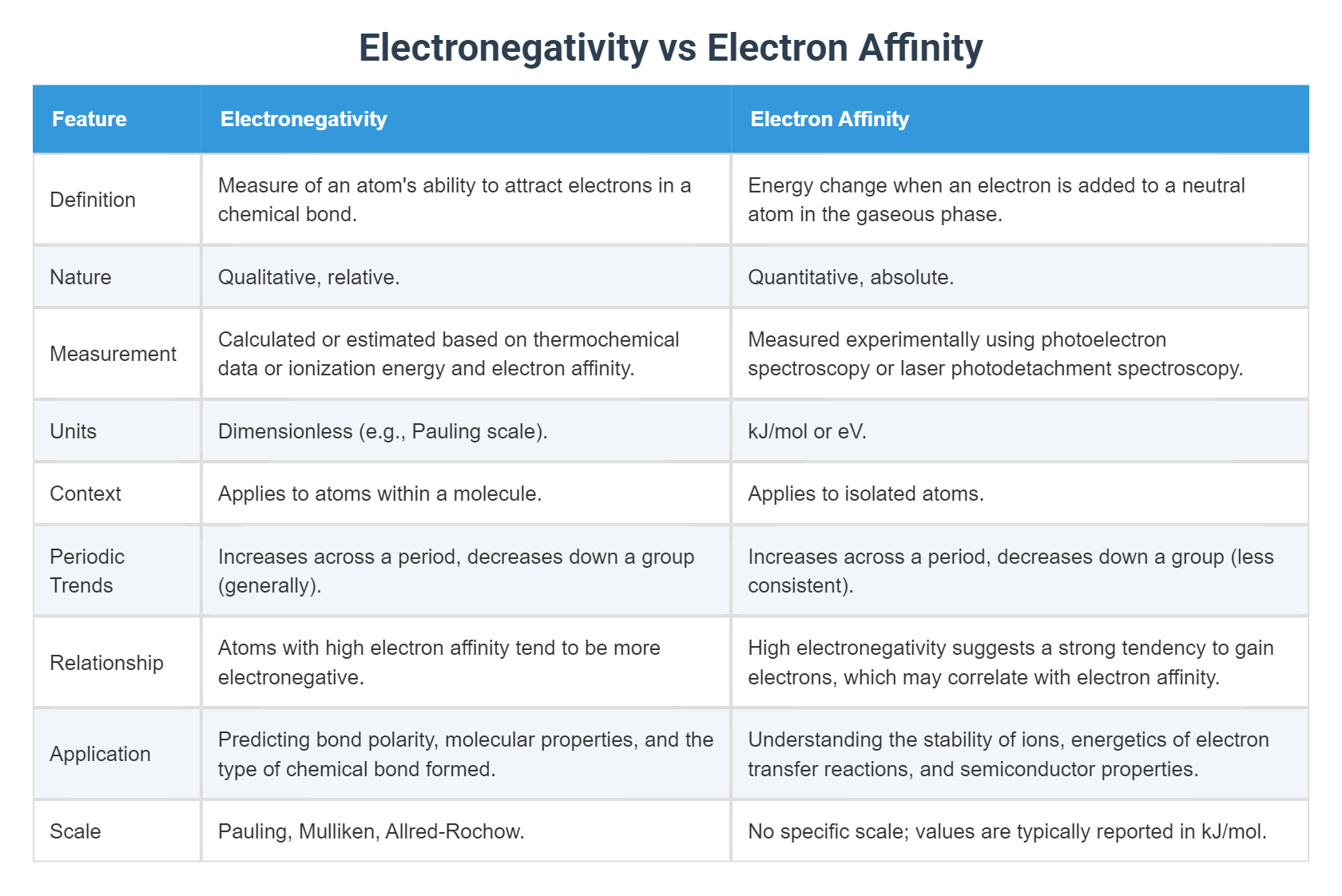
Electronegativity Vs Electron Affinity Electronegativity deals with individual atoms, while electron affinity deals with atoms in a molecule. electronegativity is also a property, while electron affinity is a measurement. Electronegativity is a chemical property that describes the ability of an atom to attract electrons towards itself in a chemical bond. while, electron affinity refers to the amount of energy released or absorbed when an electron is added to a neutral atom to form a negative ion. Electronegativity refers to the ability of an atom to attract shared electrons in a chemical bond, while electron affinity measures an atom’s tendency to accept an additional electron to form a negative ion. Electron affinity – a neutral atom can accept an electron to form a negative ion. in this process, in general, energy is released. – the electron affinity (ea) of an element is the amount of energy released when an electron is added to a gaseous atom to form an anion.
Electronegativity Vs Electron Affinity Electronegativity refers to the ability of an atom to attract shared electrons in a chemical bond, while electron affinity measures an atom’s tendency to accept an additional electron to form a negative ion. Electron affinity – a neutral atom can accept an electron to form a negative ion. in this process, in general, energy is released. – the electron affinity (ea) of an element is the amount of energy released when an electron is added to a gaseous atom to form an anion.

Electron Affinity Definition
Comments are closed.