Electronegativity Electron Affinity Lesson 4

Electronegativity And Electron Affinity Ibdp Chemistry Hl Fe2016 Focus on the valence shell and the valence electron. the key idea is that the less electron you need to take in (as compared to giving away electron), the less energy you need to use, the. Electronegativity, represented by the lowercase greek letter chi (𝛘), is the measure of the ability of an atom to attract bonding pairs of electrons. the concept of electronegativity originated because several elements have different abilities to attract electrons in a bond.

Khan Academy Electron Affinity Instructional Video For 9th 10th Electronegativity, ionization energy, and electron affinity this worksheet will cover the different trends in the periodic table. these are electronegativity, ionization energy, and electron affinity. elements behave differently because they each have a unique number of electrons. This is a crucial distinction from electron affinity: electron affinity measures the energy change when an isolated gaseous atom gains an electron, while electronegativity describes how strongly an atom pulls shared electrons toward itself when it is bonded to another atom. Recall from chapter 6 that electronegativity indicates the relative ability of an atom to attract electrons in a chemical bond. note that electronegativity values were assigned, whereas electron affinity values were measured. Electronegativity is a measure of an atom's ability to attract shared electrons to itself. on the periodic table, electronegativity generally increases as you move from left to right across a period and decreases as you move down a group.

Electron Affinity Geeksforgeeks Recall from chapter 6 that electronegativity indicates the relative ability of an atom to attract electrons in a chemical bond. note that electronegativity values were assigned, whereas electron affinity values were measured. Electronegativity is a measure of an atom's ability to attract shared electrons to itself. on the periodic table, electronegativity generally increases as you move from left to right across a period and decreases as you move down a group. Electronegativity is a measure of the ability of an atom to attract the electrons when the atom is part of a compound. electronegativity differs from electron affinity because electron affinity is the actual energy released when an atom gains an electron. Electron affinity electron affinity is the amount of energy released (or sometimes absorbed) when an atom or molecule in the gaseous state gains an electron to form a negative ion. in most cases energy is released when an electron is added because it is attracted to positive charge in the nucleus. Electron affinity is the energy change that occurs when an atom gains an electron to form a negatively charged ion (anion) in the gaseous state. it is typically expressed as a negative value since energy is released when an electron is added. The lesson is concluded with a discussion on the definition of electronegativity and a description of its periodic trend.

Electron Affinity Definition Electronegativity is a measure of the ability of an atom to attract the electrons when the atom is part of a compound. electronegativity differs from electron affinity because electron affinity is the actual energy released when an atom gains an electron. Electron affinity electron affinity is the amount of energy released (or sometimes absorbed) when an atom or molecule in the gaseous state gains an electron to form a negative ion. in most cases energy is released when an electron is added because it is attracted to positive charge in the nucleus. Electron affinity is the energy change that occurs when an atom gains an electron to form a negatively charged ion (anion) in the gaseous state. it is typically expressed as a negative value since energy is released when an electron is added. The lesson is concluded with a discussion on the definition of electronegativity and a description of its periodic trend.
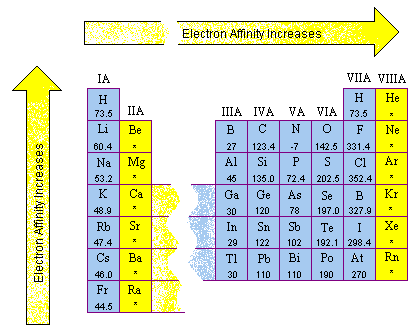
What Is Electron Affinity Socratic Electron affinity is the energy change that occurs when an atom gains an electron to form a negatively charged ion (anion) in the gaseous state. it is typically expressed as a negative value since energy is released when an electron is added. The lesson is concluded with a discussion on the definition of electronegativity and a description of its periodic trend.

Solved Electron Affinity And Electronegativity 1 Examine Chegg
Comments are closed.