Electron Affinity Trend Basic Introduction Chemistry
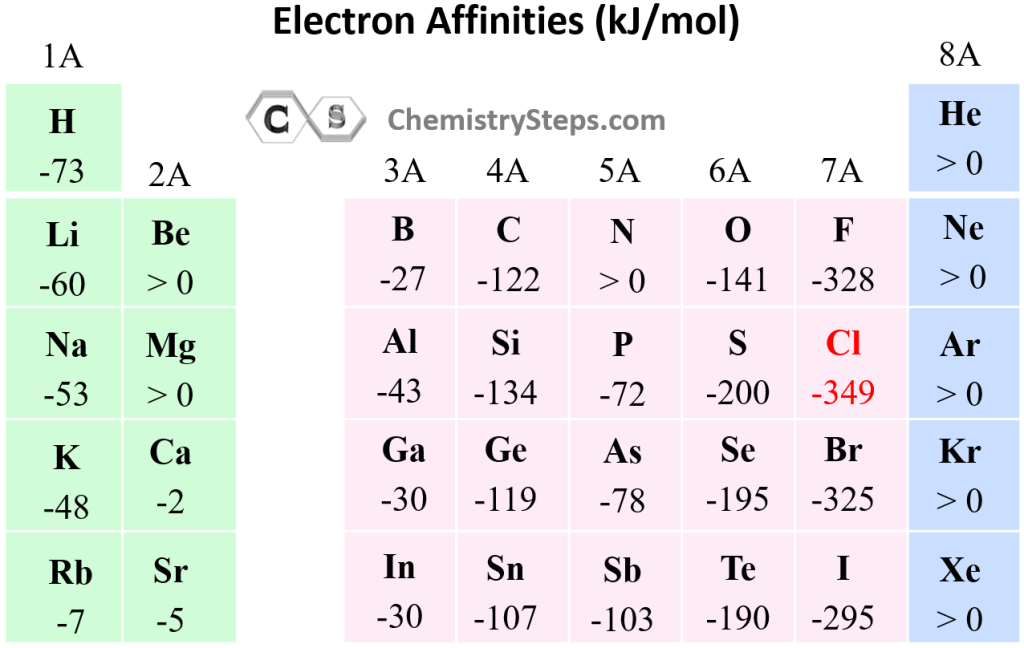
Electron Affinity Chemistry Steps This page explains electron affinity as the energy released when an atom gains an electron, typically measured in negative values in the gaseous state. it describes trends where electron affinities …. This chemistry video tutorial provides a basic introduction into electron affinity. it discusses the general trend of electron affinity as well as its many exceptions.

Periodic Trends Electron Affinity Chemistry Video Clutch Prep This study guide covers electron affinity, periodic trends, and practice questions to help you master the basics of chemistry. perfect for exam prep!. What is electron affinity. learn its equation, examples, values, and change across a period & down a group in the periodic table. also, learn its exceptions. Learn about electron affinity in chemistry. see the electron affinity trend and learn about the elements with the lowest and highest values. Learn what electron affinity means, its trends across the periodic table, real examples, exceptions, and how it differs from electronegativity for exam success.

Electron Affinity Ck 12 Foundation Learn about electron affinity in chemistry. see the electron affinity trend and learn about the elements with the lowest and highest values. Learn what electron affinity means, its trends across the periodic table, real examples, exceptions, and how it differs from electronegativity for exam success. The energy change that occurs when a neutral atom gains an electron is called its electron affinity. when energy is released in a chemical reaction or process, that energy is expressed as a negative number. Below is a visual representation of electron affinity trends throughout the periodic table. as discussed, electron affinities increase from left to right across periods; electron affinities decrease from top to bottom down groups. Electron affinity is the energy change that occurs when an electron is added to a neutral atom in the gaseous state. it measures the tendency of an atom to accept an electron. electron affinity generally increases across a period from left to right due to increasing nuclear charge. There are general trends in electron affinity across and down the periodic table of elements. electron affinity generally increases across a period in the periodic table and sometimes decreases down a group.
Comments are closed.