Electron Affinity Trend

Electron Affinity Trend Science Trends Period and group trends for electron affinities are not nearly as regular as for ionization energy. in general, electron affinities increase (become more negative) from left to right across a period and decrease (become less negative) from top to bottom down a group. Learn what electron affinity is, how it measures a neutral atom's ability to gain an electron, and how it varies on the periodic table. see examples, trends, exceptions, and the difference between electron affinity and electronegativity.
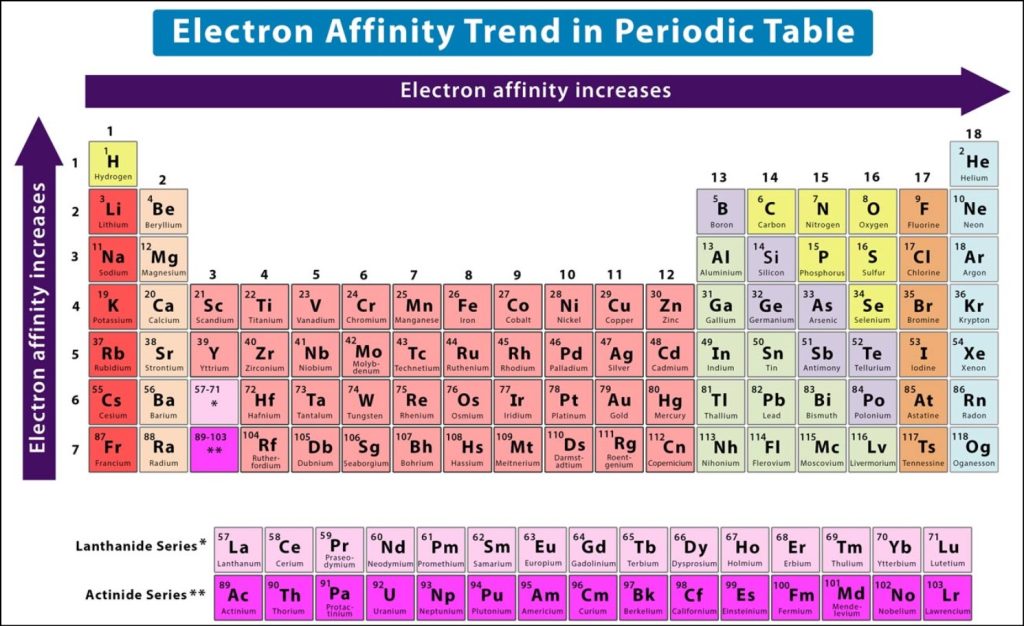
Introduction Of Electron Affinity Trend Definition Equation Learn what electron affinity is, how it differs from electronegativity and ionization energy, and how it varies across the periodic table. explore the factors that influence electron affinity, such as atomic size, nuclear charge, shielding and repulsion effects, and see examples and a chart. What is electron affinity. learn its equation, examples, values, and change across a period & down a group in the periodic table. also, learn its exceptions. In this article, we will look into what electron affinity is, its periodic trends, the factors affecting electron affinity, and the values of electron affinity of different elements. Understanding electron affinity trends across the periodic table reveals why certain elements readily form anions while others resist gaining electrons entirely.

Electron Affinity Periodic Trend Periodic Trends Periodic Table Atomic In this article, we will look into what electron affinity is, its periodic trends, the factors affecting electron affinity, and the values of electron affinity of different elements. Understanding electron affinity trends across the periodic table reveals why certain elements readily form anions while others resist gaining electrons entirely. Learn what electron affinity means, its trends across the periodic table, real examples, exceptions, and how it differs from electronegativity for exam success. Learn what electron affinity is and how it varies across the periodic table. find out the factors that affect electron affinity and the electron affinity of halogens. Electron affinity increases upward for the groups and from left to right across periods of a periodic table because the electrons added to energy levels become closer to the nucleus, thus a stronger attraction between the nucleus and its electrons. Learn how electron affinity changes across the periodic table and why it depends on the atomic structure and reactivity of elements. find examples of electron affinity values for metals and nonmetals and the factors that affect them.

Electron Affinity Periodic Trend Periodic Trends Periodic Table Atomic Learn what electron affinity means, its trends across the periodic table, real examples, exceptions, and how it differs from electronegativity for exam success. Learn what electron affinity is and how it varies across the periodic table. find out the factors that affect electron affinity and the electron affinity of halogens. Electron affinity increases upward for the groups and from left to right across periods of a periodic table because the electrons added to energy levels become closer to the nucleus, thus a stronger attraction between the nucleus and its electrons. Learn how electron affinity changes across the periodic table and why it depends on the atomic structure and reactivity of elements. find examples of electron affinity values for metals and nonmetals and the factors that affect them.
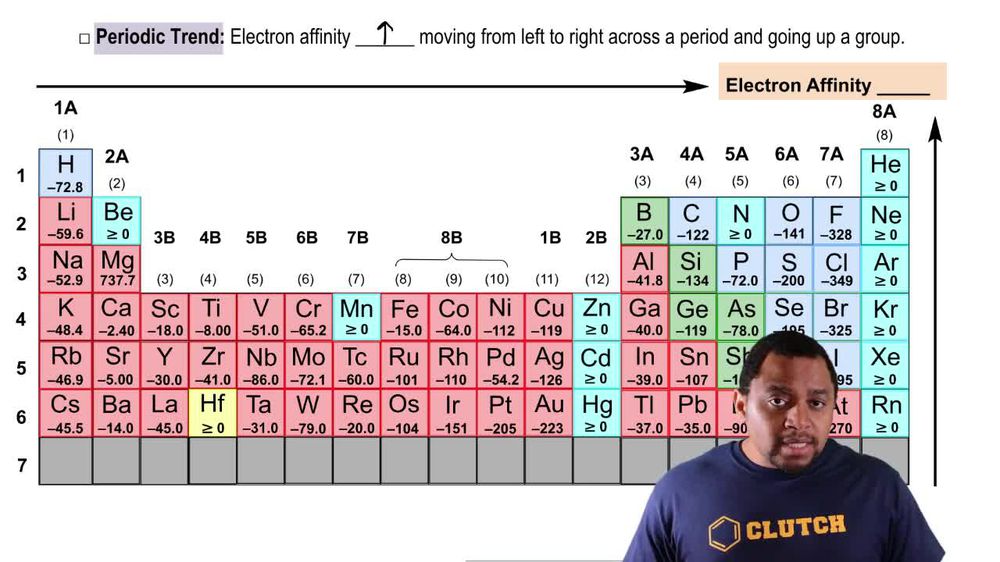
Electron Affinity Periodic Trend Periodic Trends Periodic Table Atomic Electron affinity increases upward for the groups and from left to right across periods of a periodic table because the electrons added to energy levels become closer to the nucleus, thus a stronger attraction between the nucleus and its electrons. Learn how electron affinity changes across the periodic table and why it depends on the atomic structure and reactivity of elements. find examples of electron affinity values for metals and nonmetals and the factors that affect them.
Comments are closed.