Electron Affinity Chemistry Tutorial

Electron Affinity Pdf Electron Ion This chemistry video tutorial provides a basic introduction into electron affinity. it discusses the general trend of electron affinity as well as its many exceptions. We can see that nitrogen doesn't really have an affinity for an electron, and you'll see many different values for this one, depending on which textbook you're looking in. but if we look at some electron configurations really quickly, we can try to explain this.

Electron Affinity Pdf Teaching Methods Materials Science Electron affinity is defined as the change in energy (in kj mole) of a neutral atom (in the gaseous phase) when an electron is added to the atom to form a negative ion. in other words, the neutral atom's likelihood of gaining an electron. In this article, we will look into what electron affinity is, its periodic trends, the factors affecting electron affinity, and the values of electron affinity of different elements. Learn what electron affinity means, its trends across the periodic table, real examples, exceptions, and how it differs from electronegativity for exam success. Learn what electron affinity is, how atoms gain electrons, and why electron affinity shows clear periodic trends in ib chemistry.
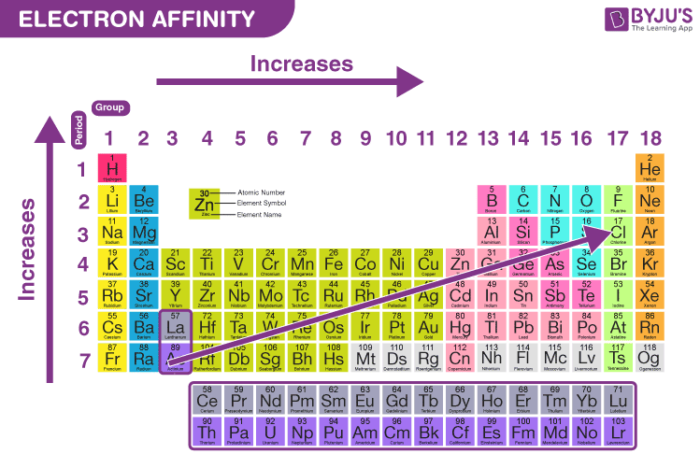
What Is Electron Affinity Definition Trends Equation With Videos Learn what electron affinity means, its trends across the periodic table, real examples, exceptions, and how it differs from electronegativity for exam success. Learn what electron affinity is, how atoms gain electrons, and why electron affinity shows clear periodic trends in ib chemistry. Periodic properties of the elements periodic trend: electron affinity. struggling with general chemistry? join thousands of students who trust us to help them ace their exams! watch the first video. This chemistry tutorial describes the concept of electron affinity and covers the general periodic trend of electron affinity along with notable exceptions in this trend. The electron affinity (ea) of an element is the energy change that occurs when an electron is added to a gaseous atom to give an anion. in general, elements with the most negative electron affinities …. Ionisation energies are always concerned with the formation of positive ions. electron affinities are the negative ion equivalent, and their use is almost always confined to elements in groups 6 and 7 of the periodic table.

Electron Affinity Chemistry Steps Periodic properties of the elements periodic trend: electron affinity. struggling with general chemistry? join thousands of students who trust us to help them ace their exams! watch the first video. This chemistry tutorial describes the concept of electron affinity and covers the general periodic trend of electron affinity along with notable exceptions in this trend. The electron affinity (ea) of an element is the energy change that occurs when an electron is added to a gaseous atom to give an anion. in general, elements with the most negative electron affinities …. Ionisation energies are always concerned with the formation of positive ions. electron affinities are the negative ion equivalent, and their use is almost always confined to elements in groups 6 and 7 of the periodic table.
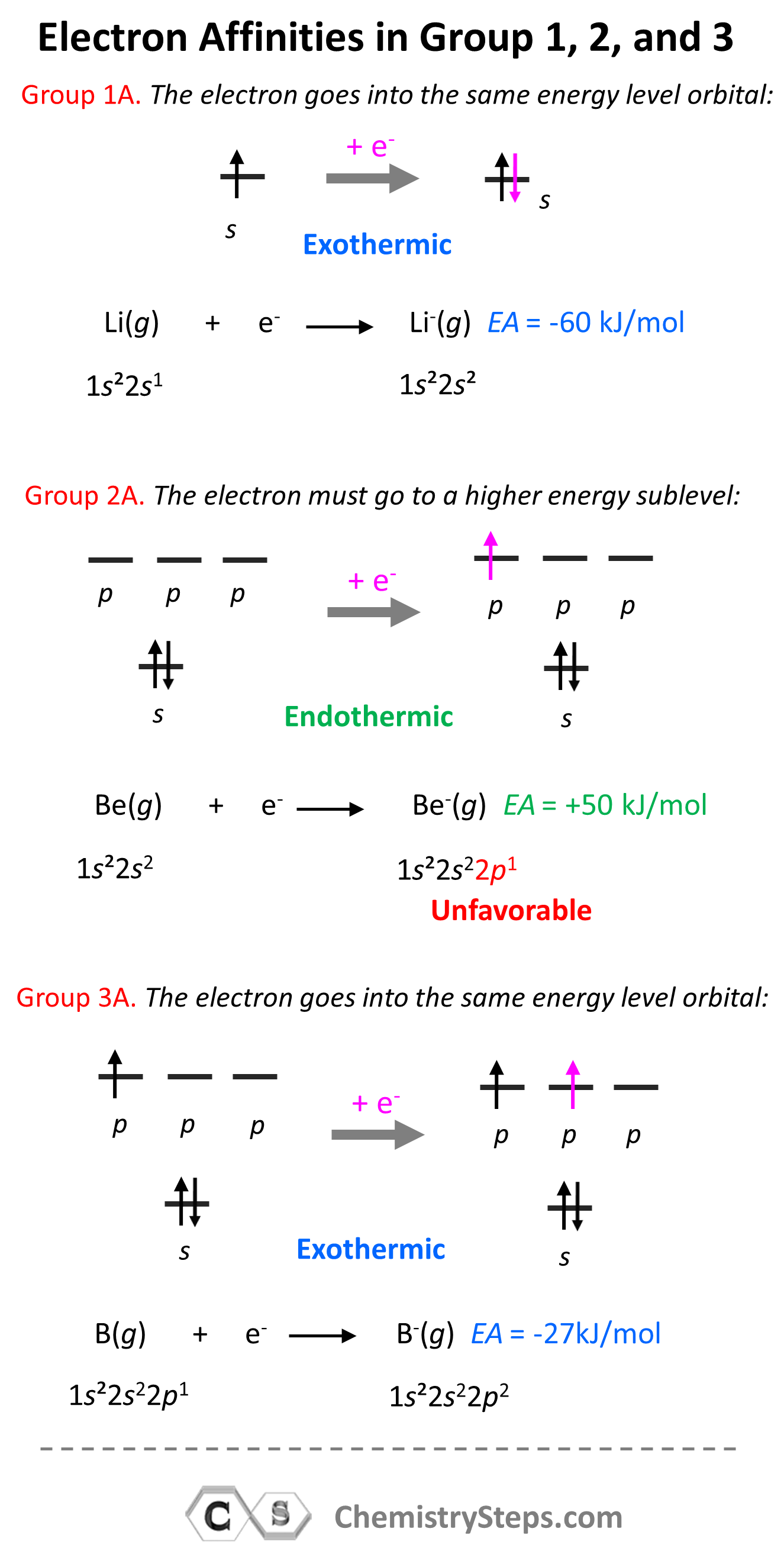
Electron Affinity Chemistry Steps The electron affinity (ea) of an element is the energy change that occurs when an electron is added to a gaseous atom to give an anion. in general, elements with the most negative electron affinities …. Ionisation energies are always concerned with the formation of positive ions. electron affinities are the negative ion equivalent, and their use is almost always confined to elements in groups 6 and 7 of the periodic table.
Comments are closed.