Electron Affinity Definition
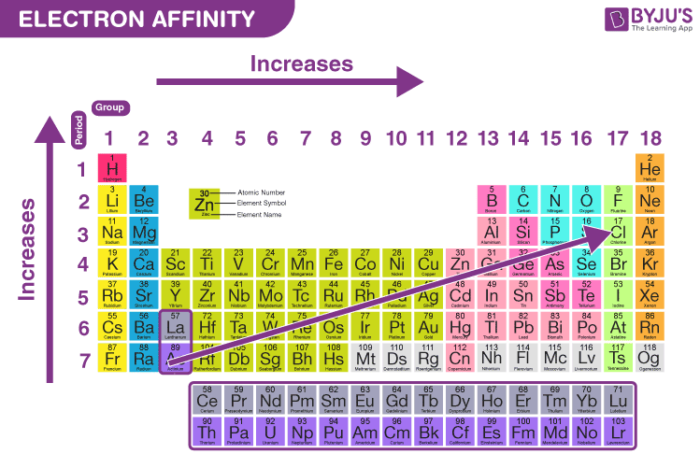
What Is Electron Affinity Definition Trends Equation With Videos Electron affinity is the energy released when an electron attaches to a neutral atom or molecule in the gaseous state. learn the definition, measurement, sign convention, and periodic table trends of electron affinity. Electron affinity is defined as the change in energy (in kj mole) of a neutral atom (in the gaseous phase) when an electron is added to the atom to form a negative ion.
.PNG)
Electron Affinity Graph Electron affinity is the energy change when a neutral atom gains an electron in the gas phase. learn how electron affinity varies across the periodic table, how it differs from electronegativity, and how to calculate it with examples. Electron affinity is the energy change that occurs when an electron is added to a neutral atom to form a negatively charged ion. it measures the attraction between the incoming electron and the nucleus, with a more negative value indicating a higher affinity for electrons. Electron affinity is the change in energy when an atom gains an electron. learn how it differs from electronegativity and ionization energy, and how it varies across the periodic table based on atomic size, nuclear charge, shielding and repulsion effects. This energy is expressed as the electron affinity, which is the energy released when an electron is attached to an atom of the element. in many cases, the electron affinity is positive, signifying that energy is indeed released when an electron attaches to an atom.

Electron Affinity Trend And Definition Electron affinity is the change in energy when an atom gains an electron. learn how it differs from electronegativity and ionization energy, and how it varies across the periodic table based on atomic size, nuclear charge, shielding and repulsion effects. This energy is expressed as the electron affinity, which is the energy released when an electron is attached to an atom of the element. in many cases, the electron affinity is positive, signifying that energy is indeed released when an electron attaches to an atom. What is electron affinity in chemistry? electron affinity refers to the energy change that occurs when an atom in the gaseous state gains an extra electron to form a negative ion. Learn what electron affinity is, how it varies across the periodic table, and how it affects chemical bonding and reactivity. find out the difference between electron affinity and electronegativity, ionization energy, and how to measure electron affinity experimentally. The equivalent more common definition is the energy released (e i n i t i a l e f i n a l) when an additional electron is attached to a neutral atom or molecule. The electron affinity is defined as the amount of energy released per mole when an electron is added to a neutral atom. it is the opposite of ionization energy [1 4].

Electron Affinity Trend And Definition What is electron affinity in chemistry? electron affinity refers to the energy change that occurs when an atom in the gaseous state gains an extra electron to form a negative ion. Learn what electron affinity is, how it varies across the periodic table, and how it affects chemical bonding and reactivity. find out the difference between electron affinity and electronegativity, ionization energy, and how to measure electron affinity experimentally. The equivalent more common definition is the energy released (e i n i t i a l e f i n a l) when an additional electron is attached to a neutral atom or molecule. The electron affinity is defined as the amount of energy released per mole when an electron is added to a neutral atom. it is the opposite of ionization energy [1 4].
Comments are closed.