Assurance Process With Ohrp
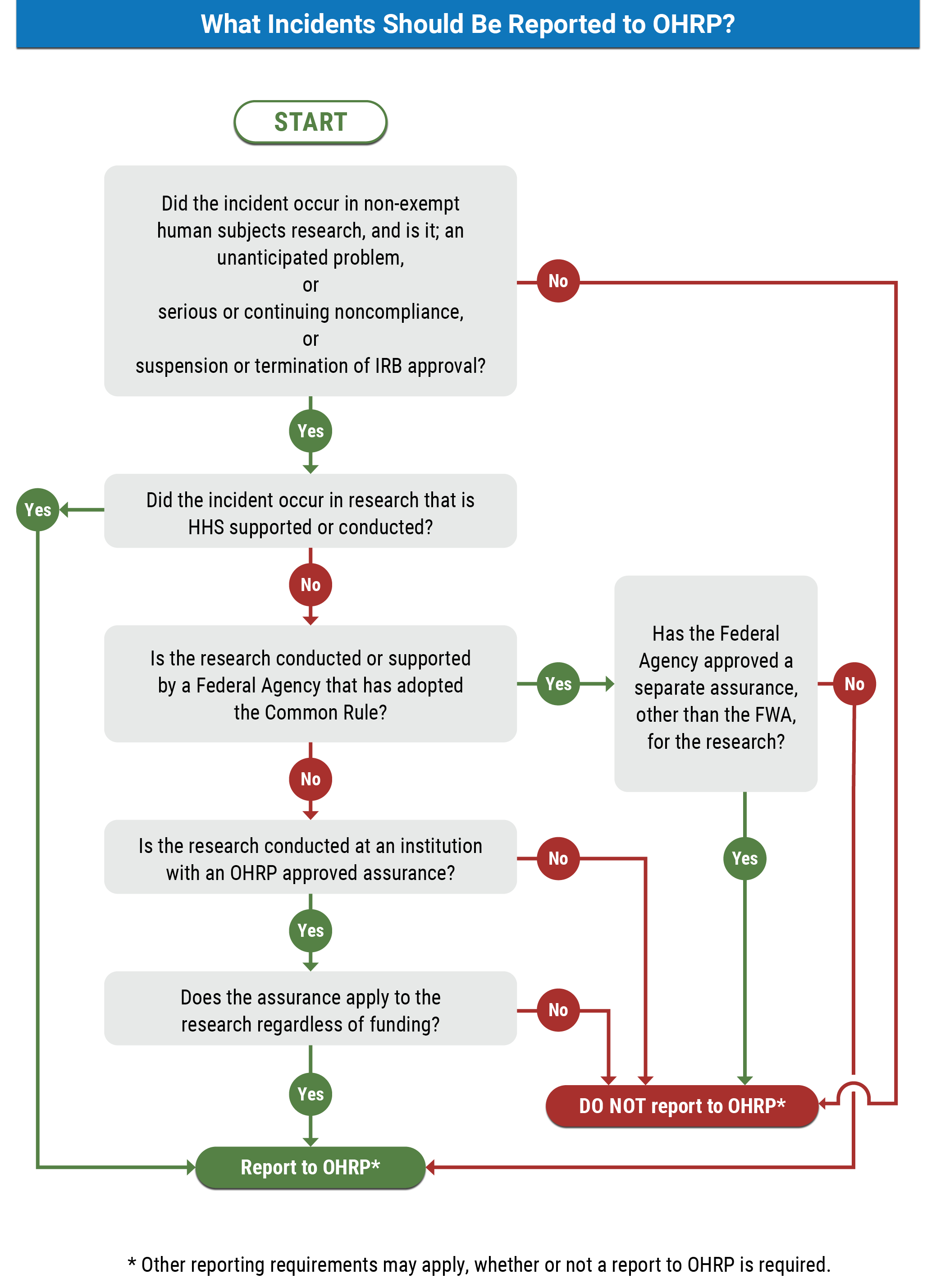
Reporting Incidents To Ohrp 2022 Hhs Gov Through the fwa and the terms of the fwa, an institution commits to hhs that it will comply with the requirements in the hhs regulations for the protection of human subjects at 45 cfr part 46. the federalwide assurance (fwa) is the only type of assurance currently accepted and approved by ohrp. The office for human research protections (ohrp) electronic submission system (ess) provides an online portal for registering institutional review boards (irbs) and obtaining federalwide assurances (fwas).

Ppt Ohrp Compliance Oversight Procedures Powerpoint Presentation The office for human research protections (ohrp) has announced significant upcoming changes to the federalwide assurance (fwa) process. these changes will streamline compliance, modernize the assurance mechanism, and align the fwa with the 2018 requirements of the common rule. A clear look at ohrp federalwide assurances — what they require, who's responsible, and how to keep your institution's fwa current and compliant. Through the assurance of compliance, an institution commits to hhs that it will comply with the requirements set forth in the regulations for the protection of human subjects at 45 cfr part 46. the federalwide assurance is the only type of assurance of compliance accepted and approved by ohrp. The university of pittsburgh holds a federal wide assurance with the office for human research protections (ohrp) in the u.s. department of health and human services (dhhs). fwas are required for every institution that is “engaged” in federally supported human subject research.

Ppt Office For Protection From Research Risks Oprr Powerpoint Through the assurance of compliance, an institution commits to hhs that it will comply with the requirements set forth in the regulations for the protection of human subjects at 45 cfr part 46. the federalwide assurance is the only type of assurance of compliance accepted and approved by ohrp. The university of pittsburgh holds a federal wide assurance with the office for human research protections (ohrp) in the u.s. department of health and human services (dhhs). fwas are required for every institution that is “engaged” in federally supported human subject research. Completing a federalwide assurance (fwa) and registering an institutional review board (irb) is a two step process. first, the irb or independent ethics committee (iec) designated under the assurance must be registered with ohrp. The office for human research protections (ohrp) has proposed updates to the federalwide assurance (fwa) form as part of its ongoing efforts to streamline regulatory processes for institutions conducting human subjects research. The federalwide assurance (fwa) is an assurance of compliance with the u.s. federal regulations for the protection of human subjects in research. it is approved by the office for human research protections (ohrp) for all human subjects research conducted or supported by the u.s. department of health and human services (hhs). An irb must be registered before it can be designated under an assurance approved for federalwide use by ohrp under § 46.103 (a). irb registration becomes effective when reviewed and accepted by ohrp.
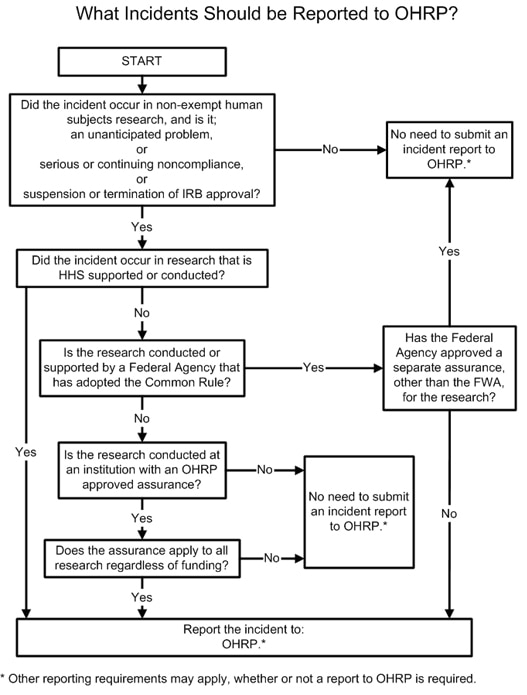
Guidance On Reporting Incidents To Ohrp 2011 Hhs Gov Completing a federalwide assurance (fwa) and registering an institutional review board (irb) is a two step process. first, the irb or independent ethics committee (iec) designated under the assurance must be registered with ohrp. The office for human research protections (ohrp) has proposed updates to the federalwide assurance (fwa) form as part of its ongoing efforts to streamline regulatory processes for institutions conducting human subjects research. The federalwide assurance (fwa) is an assurance of compliance with the u.s. federal regulations for the protection of human subjects in research. it is approved by the office for human research protections (ohrp) for all human subjects research conducted or supported by the u.s. department of health and human services (hhs). An irb must be registered before it can be designated under an assurance approved for federalwide use by ohrp under § 46.103 (a). irb registration becomes effective when reviewed and accepted by ohrp.

Ppt Reportable Events In Research Powerpoint Presentation Free The federalwide assurance (fwa) is an assurance of compliance with the u.s. federal regulations for the protection of human subjects in research. it is approved by the office for human research protections (ohrp) for all human subjects research conducted or supported by the u.s. department of health and human services (hhs). An irb must be registered before it can be designated under an assurance approved for federalwide use by ohrp under § 46.103 (a). irb registration becomes effective when reviewed and accepted by ohrp.

Ppt Research Involving Humans Ohrp Office For Human Subject
Comments are closed.