When The Assurance Comes A Knockin Ohrps Fwa And Irb Registration Processes

Irb Fwa Institutional Review Board Learn the status of an irb registration or a fwa submission. find a variety of forms (e.g., sample irb authorization, individual investigator agreements, irb registration and fwa instructions, and terms of assurances). contact ohrp with questions about fwas or irb registration. The office for human research protections (ohrp) electronic submission system (ess) provides an online portal for registering institutional review boards (irbs) and obtaining federalwide assurances (fwas).

Irb Fwa Institutional Review Board These changes will streamline compliance, modernize the assurance mechanism, and align the fwa with the 2018 requirements of the common rule. these updates will also temporarily affect access to ohrp’s electronic submission system for both fwas and institutional review board (irb) registrations. This webinar will feature dr. irene stith coleman, dr. hal blatt, and ms. jean makle with the division of policy and assurances, ohrp. they will discuss the requirements and processes. A clear look at ohrp federalwide assurances — what they require, who's responsible, and how to keep your institution's fwa current and compliant. Watch educational videos the videos below cover material that is particularly relevant to irb members • ohrp reporting requirements • irb records, parts i and ii • when the assurance comes a knockin': ohrp's fwa and irb registration process • when the feds come a knockin': how to prepare for an ohrp compliance evaluation of your program.

Fwa Irb Registrations Ucla Office Of The Human Research Protection A clear look at ohrp federalwide assurances — what they require, who's responsible, and how to keep your institution's fwa current and compliant. Watch educational videos the videos below cover material that is particularly relevant to irb members • ohrp reporting requirements • irb records, parts i and ii • when the assurance comes a knockin': ohrp's fwa and irb registration process • when the feds come a knockin': how to prepare for an ohrp compliance evaluation of your program. Here an institution will find information about when the fwa was received and which ohrp assurance coordinator is reviewing its application and how to contact that person. Watch these instructional videos to help you file or renew your fwa smoothly. learn how to file a new fwa with ohrp. through the fwa and the terms of the fwa, an institution commits to hhs that it will comply with the requirements in the hhs protection of human subjects regulations at 45 cfr part 46. What is an assurance of compliance with human subject protection regulations? an assurance of compliance is a written document submitted by an institution (not an institutional review board) that is engaged in non exempt human subjects research conducted or supported by hhs. Learn how to register an irb with ohrp online. update an irb registration within 90 days after a change in contact person or chairperson, and renew irb registration every 3 years. access faqs about the irb registration process. irb organizations (iorgs) can register one or more irb (s).

Chiper Irb Registration And Fwa Compliance Reminder Research Manitoba Here an institution will find information about when the fwa was received and which ohrp assurance coordinator is reviewing its application and how to contact that person. Watch these instructional videos to help you file or renew your fwa smoothly. learn how to file a new fwa with ohrp. through the fwa and the terms of the fwa, an institution commits to hhs that it will comply with the requirements in the hhs protection of human subjects regulations at 45 cfr part 46. What is an assurance of compliance with human subject protection regulations? an assurance of compliance is a written document submitted by an institution (not an institutional review board) that is engaged in non exempt human subjects research conducted or supported by hhs. Learn how to register an irb with ohrp online. update an irb registration within 90 days after a change in contact person or chairperson, and renew irb registration every 3 years. access faqs about the irb registration process. irb organizations (iorgs) can register one or more irb (s).
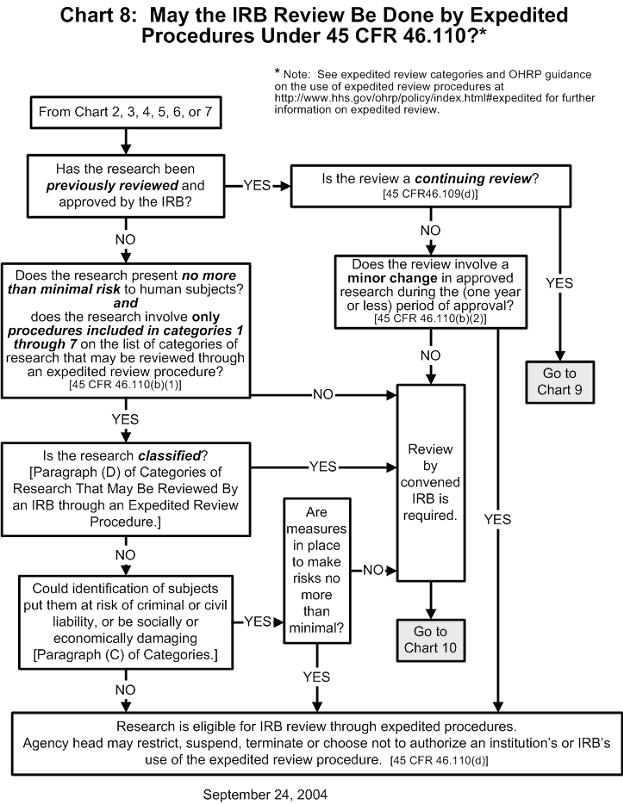
Documents What is an assurance of compliance with human subject protection regulations? an assurance of compliance is a written document submitted by an institution (not an institutional review board) that is engaged in non exempt human subjects research conducted or supported by hhs. Learn how to register an irb with ohrp online. update an irb registration within 90 days after a change in contact person or chairperson, and renew irb registration every 3 years. access faqs about the irb registration process. irb organizations (iorgs) can register one or more irb (s).
Comments are closed.