Breaking Down Ohrp Assurances
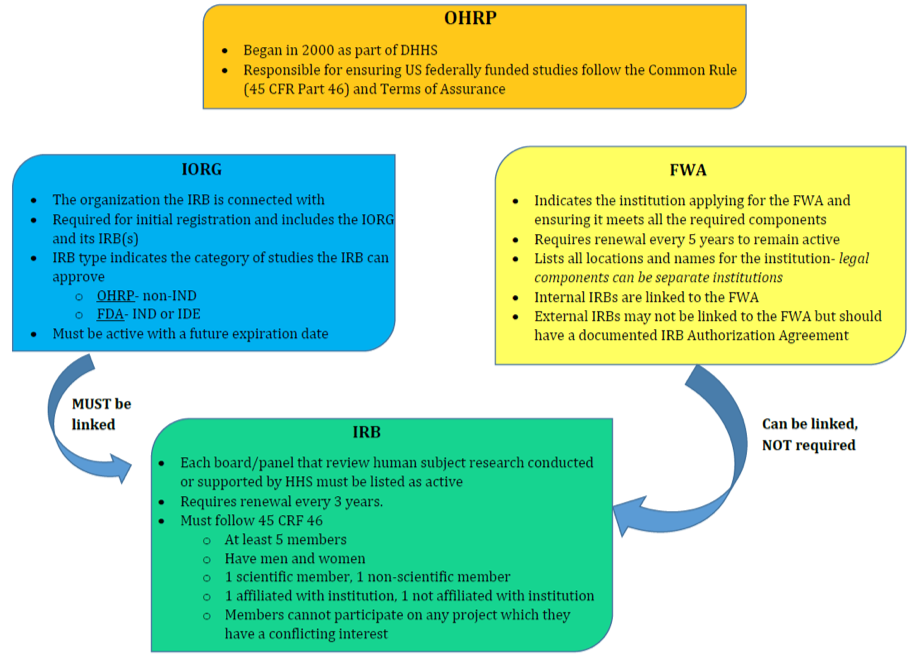
Breaking Down Ohrp Assurances In particular, this paper will focus on specific requirements to ensure the protection of human research subjects (title 45 cfr 46). Through the assurance of compliance, an institution commits to hhs that it will comply with the requirements set forth in the regulations for the protection of human subjects at 45 cfr part 46. the federalwide assurance is the only type of assurance of compliance accepted and approved by ohrp.
.png)
Breaking Down Ohrp Assurances Applied Clinical Trials Online Therefore, an institution (domestically and internationally) engaged in nonexempt human subject’s research conducted or supported by federal funds must submit to ohrp written assurance that it will comply with the hhs regulations, known as a federal wide assurance (fwa). Imagine a massive, nationwide clinical trial is testing a promising new drug. thousands of volunteers—your neighbors, friends, maybe even you—agree to participate, trusting that their safety and rights are the top priority. who ensures that trust isn't broken?. To promote efficiency and accountability in both government activity and government sponsored research, ohrp must have the experienced leadership, resources, and expertise necessary to:. Through the fwa and the terms of the fwa, an institution commits to hhs that it will comply with the requirements in the hhs regulations for the protection of human subjects at 45 cfr part 46. the federalwide assurance (fwa) is the only type of assurance currently accepted and approved by ohrp.

Breaking Down Ohrp Assurances Applied Clinical Trials Online To promote efficiency and accountability in both government activity and government sponsored research, ohrp must have the experienced leadership, resources, and expertise necessary to:. Through the fwa and the terms of the fwa, an institution commits to hhs that it will comply with the requirements in the hhs regulations for the protection of human subjects at 45 cfr part 46. the federalwide assurance (fwa) is the only type of assurance currently accepted and approved by ohrp. The federalwide assurance (fwa) is an assurance of compliance with the u.s. federal regulations for the protection of human subjects in research. it is approved by the office for human research protections (ohrp) for all human subjects research conducted or supported by the u.s. department of health and human services (hhs). To avoid delays in the ohro approval process, verify that the institutions engaged in the research have active assurances. the institution’s irb office or the ohro can assist in determining if engaged institutions have active assurances and obtaining an assurance is required. Any renewal or update that is submitted to, and accepted by, ohrp begins a new 5 year effective period. failure to renew or update an fwa appropriately may result in restriction, suspension, or termination of ohrp’s approval of the institution’s fwa. The office for human research protections (ohrp) provides leadership in the protection of the rights, welfare, and wellbeing of subjects involved in research conducted or supported by the u.s. department of health and human services.
Breaking Down Ohrp Assurances Applied Clinical Trials Online The federalwide assurance (fwa) is an assurance of compliance with the u.s. federal regulations for the protection of human subjects in research. it is approved by the office for human research protections (ohrp) for all human subjects research conducted or supported by the u.s. department of health and human services (hhs). To avoid delays in the ohro approval process, verify that the institutions engaged in the research have active assurances. the institution’s irb office or the ohro can assist in determining if engaged institutions have active assurances and obtaining an assurance is required. Any renewal or update that is submitted to, and accepted by, ohrp begins a new 5 year effective period. failure to renew or update an fwa appropriately may result in restriction, suspension, or termination of ohrp’s approval of the institution’s fwa. The office for human research protections (ohrp) provides leadership in the protection of the rights, welfare, and wellbeing of subjects involved in research conducted or supported by the u.s. department of health and human services.
Comments are closed.