Ohrp Irb Records Part One

Irb Guidebook Scope: this guidance document applies to nonexempt human subjects research activities that are conducted or supported by hhs. it applies to cooperative research projects that involve more than one institution for which the 2018 requirements, including the single irb review requirement, apply. In this office for human research protections (ohrp) video, all of the characters are fictional. this video provides information regarding the irb records requirements described at 45 cfr part 46.

Irb Hhs Gov The database of registered iorgs and irbs includes information on irbs that are regulated by ohrp only, ohrp and fda , and fda only. Note: this video was created before the 2018 revisions of the common rule and may include information that is not up to date.in this office for human researc. The institution or irb may maintain the records in printed form, or electronically. all records shall be accessible for inspection and copying by authorized representatives of the federal department or agency at reasonable times and in a reasonable manner. Supplementary information: i. background ohrp is announcing the availability of a draft guidance document for public a single institutional re ly for institutio review boards (irbs), investigators, institutional officials, and other human research protection staff.
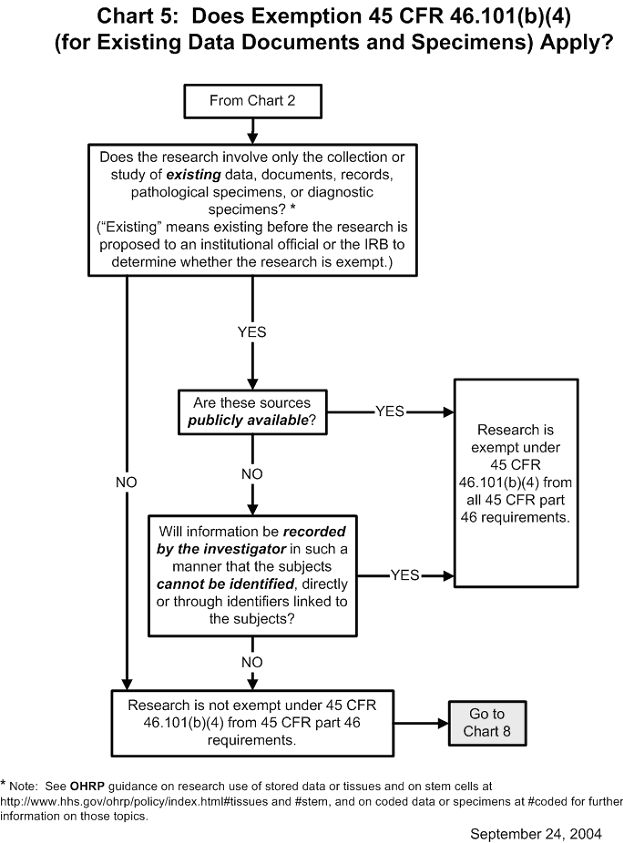
Documents The institution or irb may maintain the records in printed form, or electronically. all records shall be accessible for inspection and copying by authorized representatives of the federal department or agency at reasonable times and in a reasonable manner. Supplementary information: i. background ohrp is announcing the availability of a draft guidance document for public a single institutional re ly for institutio review boards (irbs), investigators, institutional officials, and other human research protection staff. Each institutional review board (irb) that reviews research involving human subjects conducted or supported by the department of health and human services (hhs) must be registered with the office for human research protections (ohrp). Ohrp and fda believe that when institutions and irbs develop and follow clear written procedures, there is an increased likelihood that the rights and welfare of human subjects will be. An irb must be registered before it can be designated under an assurance approved for federalwide use by ohrp under § 46.103 (a). irb registration becomes effective when reviewed and accepted by ohrp. Learn how institutions can register irbs and obtain fwas. find out more about the secretary’s advisory committee on human research protections. learn how ohrp promotes ethical conduct and regulatory compliance in hhs supported research studies conducted outside the united states.
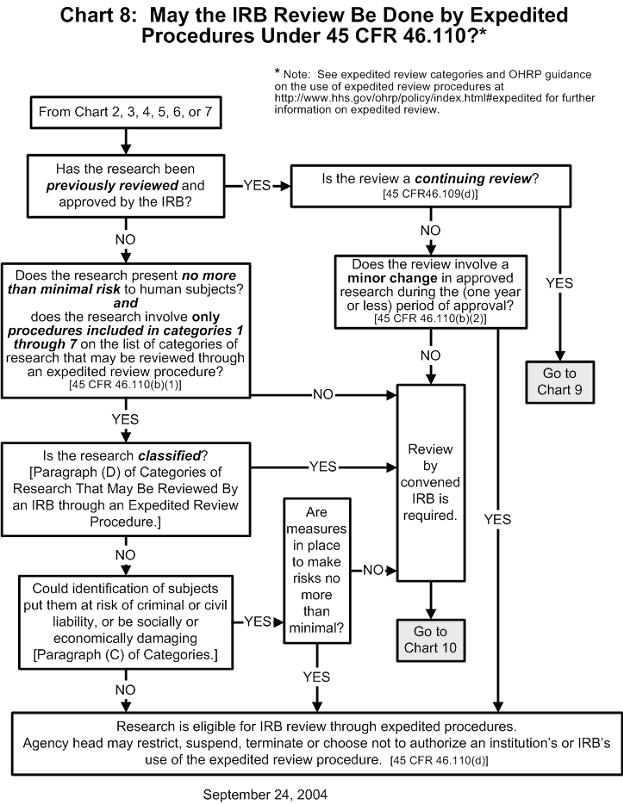
Documents Each institutional review board (irb) that reviews research involving human subjects conducted or supported by the department of health and human services (hhs) must be registered with the office for human research protections (ohrp). Ohrp and fda believe that when institutions and irbs develop and follow clear written procedures, there is an increased likelihood that the rights and welfare of human subjects will be. An irb must be registered before it can be designated under an assurance approved for federalwide use by ohrp under § 46.103 (a). irb registration becomes effective when reviewed and accepted by ohrp. Learn how institutions can register irbs and obtain fwas. find out more about the secretary’s advisory committee on human research protections. learn how ohrp promotes ethical conduct and regulatory compliance in hhs supported research studies conducted outside the united states.
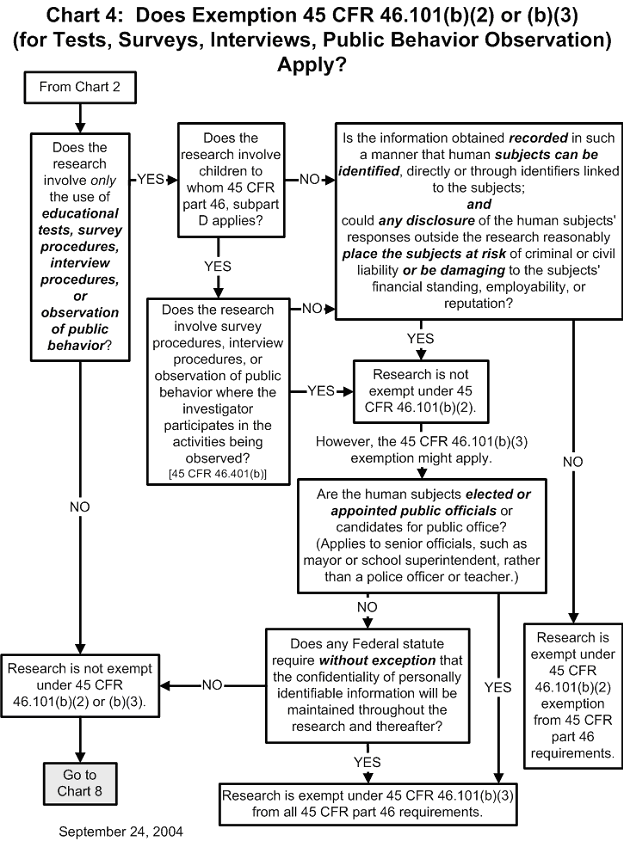
Documents An irb must be registered before it can be designated under an assurance approved for federalwide use by ohrp under § 46.103 (a). irb registration becomes effective when reviewed and accepted by ohrp. Learn how institutions can register irbs and obtain fwas. find out more about the secretary’s advisory committee on human research protections. learn how ohrp promotes ethical conduct and regulatory compliance in hhs supported research studies conducted outside the united states.
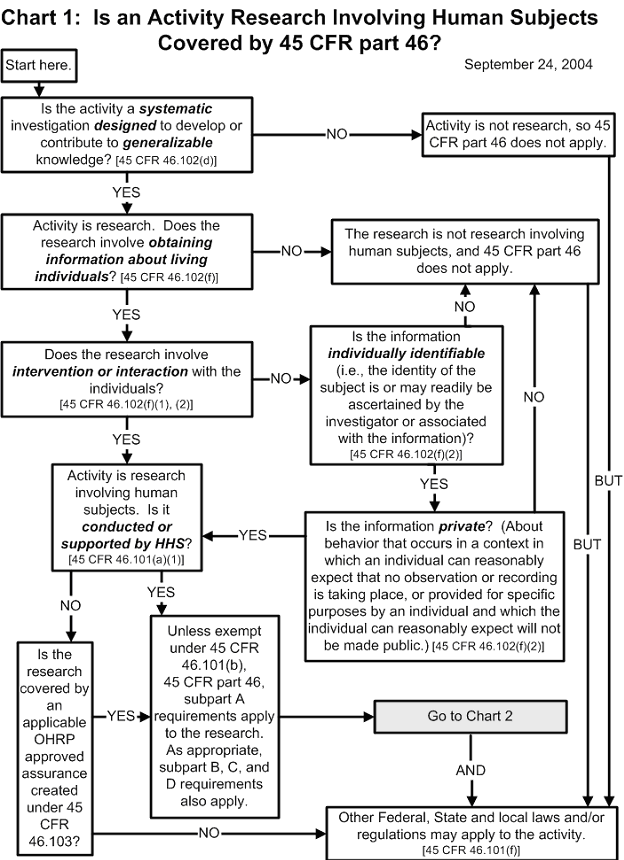
Documents
Comments are closed.