Ohrp Irb Records Part Two

Irb Hhs Gov In this office for human research protections (ohrp) video, all of the characters are fictional. this video provides information regarding the irb records requirements described at 45 cfr. Ohrp educational videos were developed by the ohrp division of education and development and are intended to provide information regarding the requirements of the department of health and human services (hhs) regulations for the protection of human subjects at 45 cfr part 46.
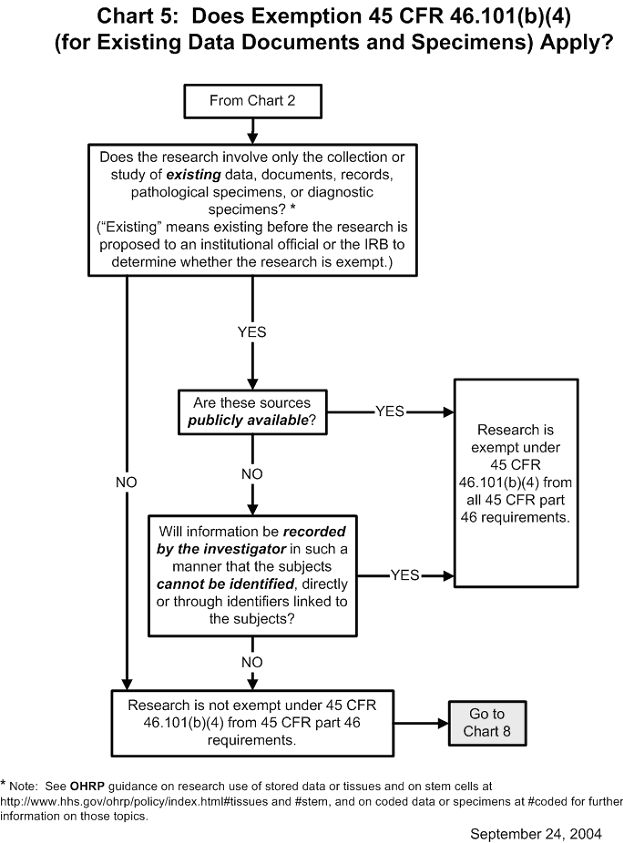
Documents Each institutional review board (irb) that reviews research involving human subjects conducted or supported by the department of health and human services (hhs) must be registered with the office for human research protections (ohrp). The database of registered iorgs and irbs includes information on irbs that are regulated by ohrp only, ohrp and fda , and fda only. (b) the records required by this policy shall be retained for at least 3 years, and records relating to research that is conducted shall be retained for at least 3 years after completion of the research. the institution or irb may maintain the records in printed form, or electronically. Although the subpart d regulations do not explicitly require an irb to follow a written procedure for review of studies involving children, ohrp and fda recommend that written procedures for.
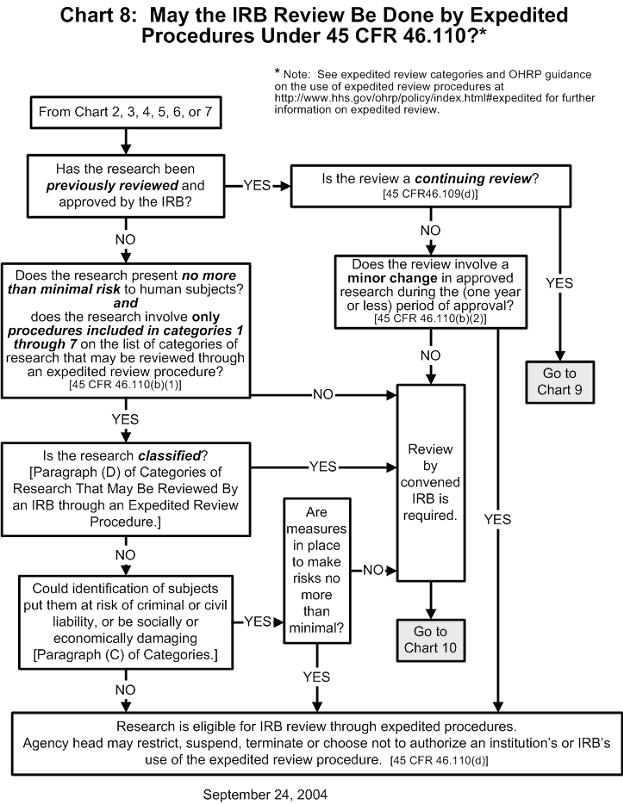
Documents (b) the records required by this policy shall be retained for at least 3 years, and records relating to research that is conducted shall be retained for at least 3 years after completion of the research. the institution or irb may maintain the records in printed form, or electronically. Although the subpart d regulations do not explicitly require an irb to follow a written procedure for review of studies involving children, ohrp and fda recommend that written procedures for. The office for human research protections (ohrp) provides leadership in the protection of the rights, welfare, and wellbeing of human subjects involved in research conducted or supported by the u.s. department of health and human services (hhs). Agreements with institutional review boards (irb) or between institutions organizations and irbs are not the same as fwas. irb agreements do not substitute for or replace fwas. Ohrp training resources on human research protection by u.s. department of health and human services •. Choosing an item from citations and headings will bring you directly to the content. choosing an item from full text search results will bring you to those results. pressing enter in the search box will also bring you to search results. these links go to the official, published cfr, which is updated annually.
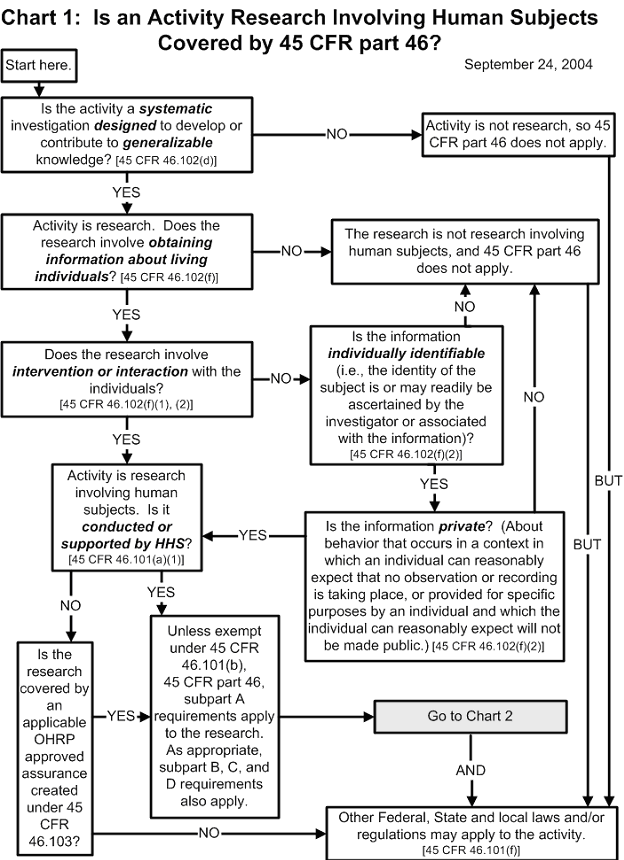
Documents The office for human research protections (ohrp) provides leadership in the protection of the rights, welfare, and wellbeing of human subjects involved in research conducted or supported by the u.s. department of health and human services (hhs). Agreements with institutional review boards (irb) or between institutions organizations and irbs are not the same as fwas. irb agreements do not substitute for or replace fwas. Ohrp training resources on human research protection by u.s. department of health and human services •. Choosing an item from citations and headings will bring you directly to the content. choosing an item from full text search results will bring you to those results. pressing enter in the search box will also bring you to search results. these links go to the official, published cfr, which is updated annually.
Comments are closed.