Redox Reaction1

Reduction Oxidation Reaction Redox Reaction Biorender Science Templates What is a redox reaction? a redox reaction is a chemical reaction in which the atoms change their oxidation numbers. some atoms lose electrons and are oxidized – a process known as oxidation. on the other hand, some atoms gain electrons and are reduced – a process known as reduction. An oxidation reduction (redox) reaction is a type of chemical reaction that involves a transfer of electrons between two species. an oxidation reduction reaction is any chemical reaction in which the oxidation number of a molecule, atom, or ion changes by gaining or losing an electron.

Redox Reaction Redox Reactions Oxidation And Reduction O Level Redox reactions can occur slowly, as in the formation of rust, or rapidly, as in the case of burning fuel. electron transfer reactions are generally fast, occurring within the time of mixing. Redox reactions involve the transfer of electrons between reactant species and are distinct from other reaction types, such as acid base, which involve the transfer of protons. oxidation is the loss of electron (s), and reduction is the gain of electron (s). This tutorial covers oxidation reduction (redox) reactions in electrochemistry: learn to write simple half reactions and to recognize the oxidizing agent and reducing agent. An oxidation reduction reaction is any chemical reaction in which the oxidation number of a participating chemical species changes. many such reactions are common and familiar—combustion, rusting, cellular respiration, and photosynthesis are some examples.

Redox Reaction This tutorial covers oxidation reduction (redox) reactions in electrochemistry: learn to write simple half reactions and to recognize the oxidizing agent and reducing agent. An oxidation reduction reaction is any chemical reaction in which the oxidation number of a participating chemical species changes. many such reactions are common and familiar—combustion, rusting, cellular respiration, and photosynthesis are some examples. A redox reaction, short for reduction–oxidation reaction, is a chemical process in which one substance loses electrons (oxidation) while another gains electrons (reduction). In this tutorial, you will learn what a redox reaction is, the different parts of such a reaction, as well as how to recognize and write redox reactions. you will also learn the difference between oxidation and reduction, and the definition of oxidation. Redox reactions are defined as chemical reactions in which the acquisition and loss of electrons occur, often leading to changes in electron density and other properties of the involved substances. Redox reactions play a critical role in the functioning of batteries as they consist of two electrodes, an anode and a cathode, separated by an electrolyte. when the battery is discharging, a chemical reaction occurs at the anode (oxidation) and another at the cathode (reduction).
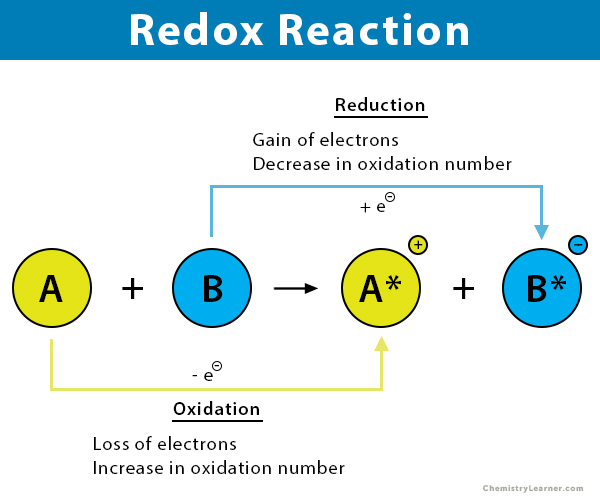
Redox Reaction Meaning A redox reaction, short for reduction–oxidation reaction, is a chemical process in which one substance loses electrons (oxidation) while another gains electrons (reduction). In this tutorial, you will learn what a redox reaction is, the different parts of such a reaction, as well as how to recognize and write redox reactions. you will also learn the difference between oxidation and reduction, and the definition of oxidation. Redox reactions are defined as chemical reactions in which the acquisition and loss of electrons occur, often leading to changes in electron density and other properties of the involved substances. Redox reactions play a critical role in the functioning of batteries as they consist of two electrodes, an anode and a cathode, separated by an electrolyte. when the battery is discharging, a chemical reaction occurs at the anode (oxidation) and another at the cathode (reduction).
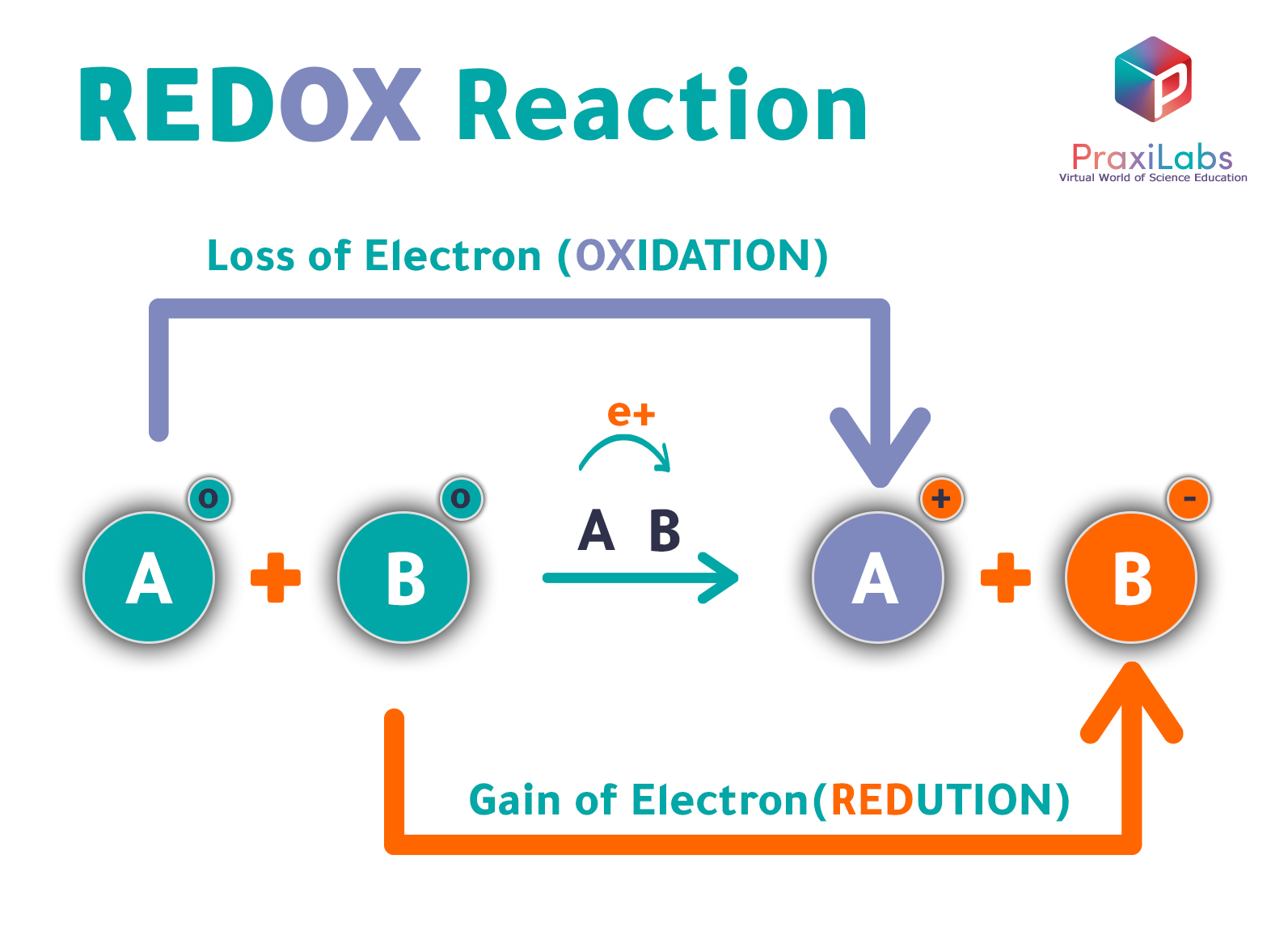
Oxidation And Reduction Reactions With Examples Praxilabs Redox reactions are defined as chemical reactions in which the acquisition and loss of electrons occur, often leading to changes in electron density and other properties of the involved substances. Redox reactions play a critical role in the functioning of batteries as they consist of two electrodes, an anode and a cathode, separated by an electrolyte. when the battery is discharging, a chemical reaction occurs at the anode (oxidation) and another at the cathode (reduction).
Comments are closed.