Redox Reaction Meaning

Reduction Oxidation Reaction Redox Reaction Biorender Science Templates What is a redox reaction? a redox reaction is a chemical reaction in which the atoms change their oxidation numbers. some atoms lose electrons and are oxidized – a process known as oxidation. on the other hand, some atoms gain electrons and are reduced – a process known as reduction. Redox is a type of chemical reaction in which the oxidation states of the reactants change. oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a decrease in the oxidation state. learn more about redox reactions, electron transfer and atom transfer processes, and related terms.
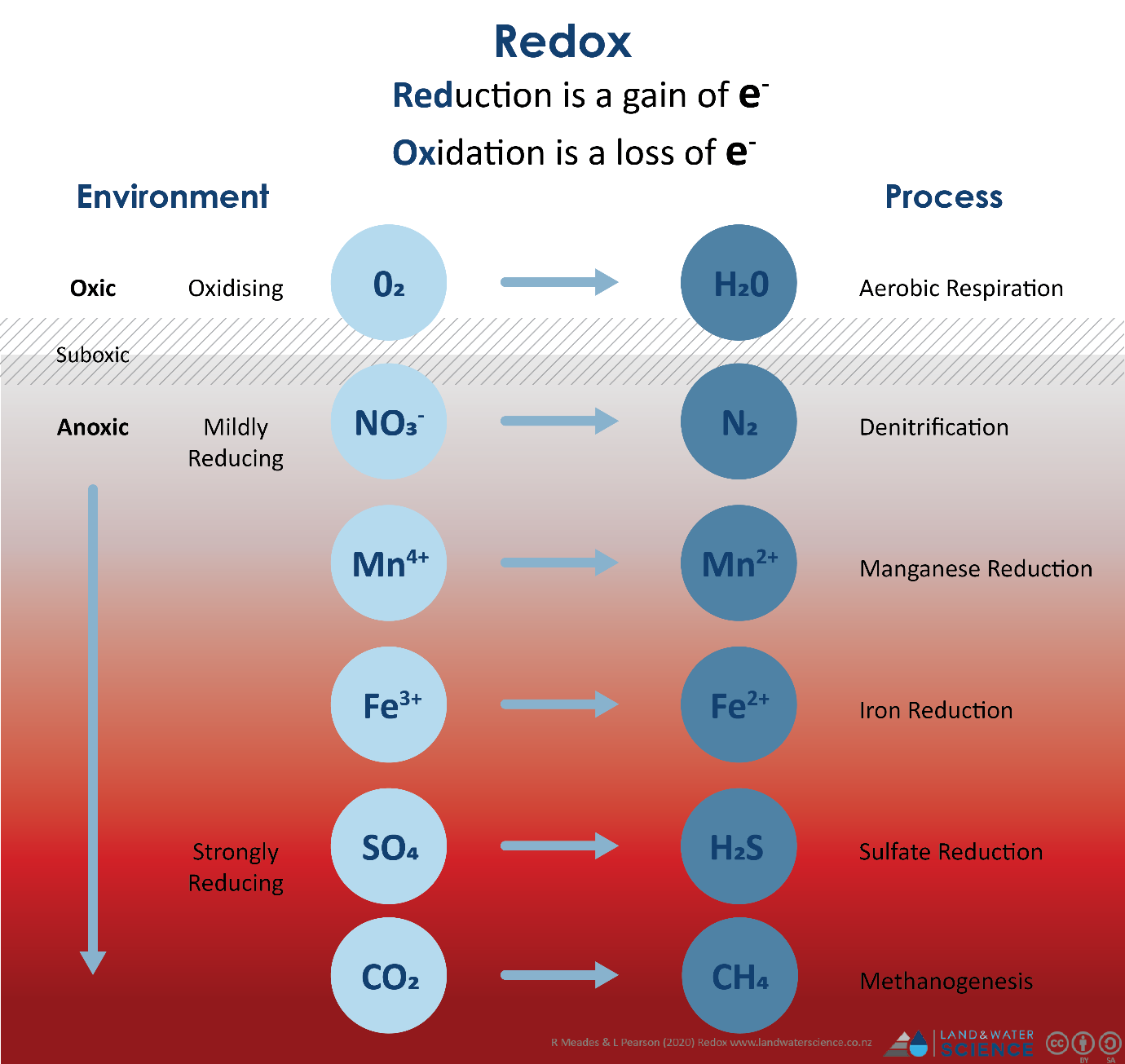
Redox Reaction Redox Reactions Oxidation And Reduction O Level An oxidation reduction (redox) reaction is a type of chemical reaction that involves a transfer of electrons between two species. an oxidation reduction reaction is any chemical reaction in which the oxidation number of a molecule, atom, or ion changes by gaining or losing an electron. Because of their complementary nature, the oxidation and reduction processes together are referred to as redox reactions. the reactant that brings about the oxidation is called the oxidizing agent, and that reagent is itself reduced by the reducing agent. Redox reactions (oxidation–reduction reactions) are characterized as electron transfer reactions. like acid–base reactions, oxidants accept protons and reductants donate electrons. If the oxidation number of any element changes during the reaction, it is a redox reaction. an increase in oxidation number indicates oxidation, while a decrease indicates reduction.
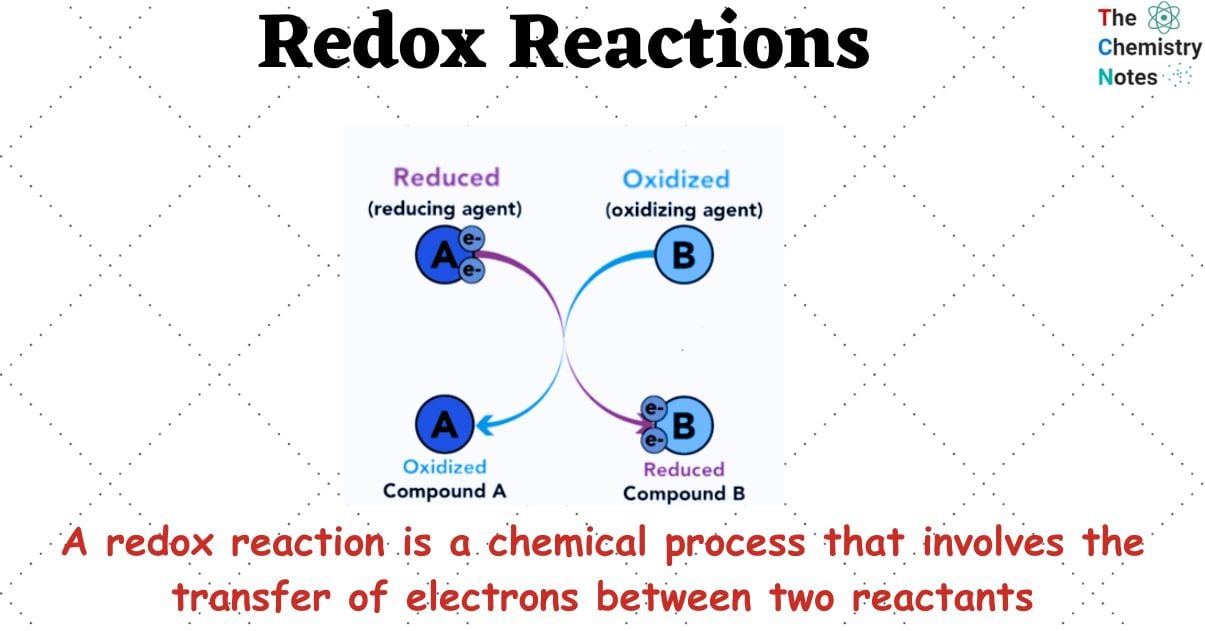
Redox Reaction Redox Reactions Oxidation And Reduction O Level Redox reactions (oxidation–reduction reactions) are characterized as electron transfer reactions. like acid–base reactions, oxidants accept protons and reductants donate electrons. If the oxidation number of any element changes during the reaction, it is a redox reaction. an increase in oxidation number indicates oxidation, while a decrease indicates reduction. Learn what a redox reaction is, how to recognize and write it, and the difference between oxidation and reduction. see examples of redox reactions involving metals, nonmetals, oxygen, and bleach. A redox reaction, short for reduction–oxidation reaction, is a chemical process in which one substance loses electrons (oxidation) while another gains electrons (reduction). What does redox mean in simple terms? redox is a chemical reaction that involves the transfer of electrons. the word “redox” is made up of two words: reduction and oxidation. reduction happens when something loses electrons. oxidation happens when something gains electrons. Redox reactions are the foundation of electrode and cell potentials in electrochemical systems. when two different metals or electrodes are placed in an electrolyte solution, a potential difference arises due to the tendency of one metal to lose electrons (oxidation) while the other gains electrons (reduction).
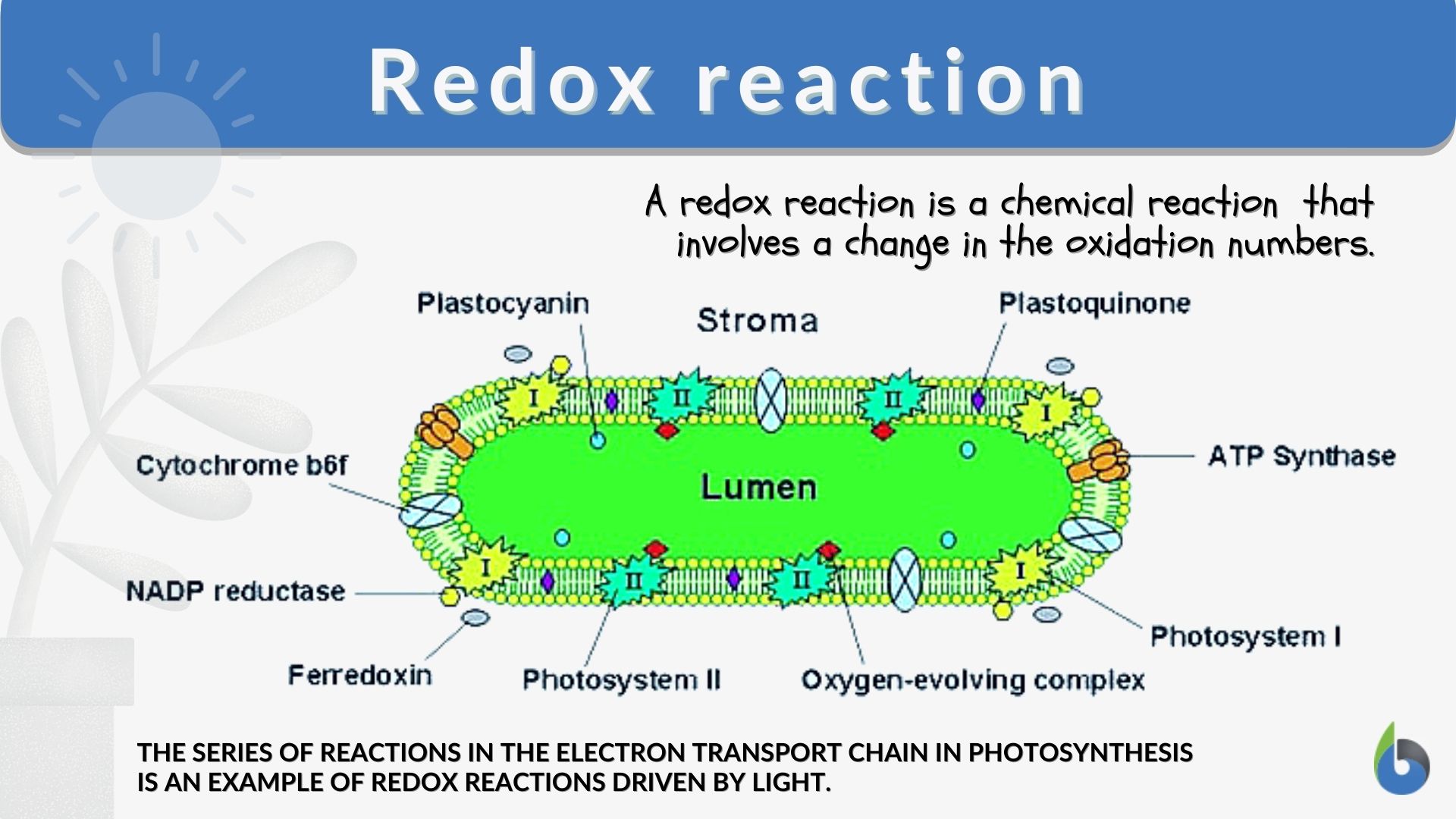
Redox Reaction Definition And Examples Biology Online Dictionary Learn what a redox reaction is, how to recognize and write it, and the difference between oxidation and reduction. see examples of redox reactions involving metals, nonmetals, oxygen, and bleach. A redox reaction, short for reduction–oxidation reaction, is a chemical process in which one substance loses electrons (oxidation) while another gains electrons (reduction). What does redox mean in simple terms? redox is a chemical reaction that involves the transfer of electrons. the word “redox” is made up of two words: reduction and oxidation. reduction happens when something loses electrons. oxidation happens when something gains electrons. Redox reactions are the foundation of electrode and cell potentials in electrochemical systems. when two different metals or electrodes are placed in an electrolyte solution, a potential difference arises due to the tendency of one metal to lose electrons (oxidation) while the other gains electrons (reduction).
Comments are closed.