Reduction Reaction
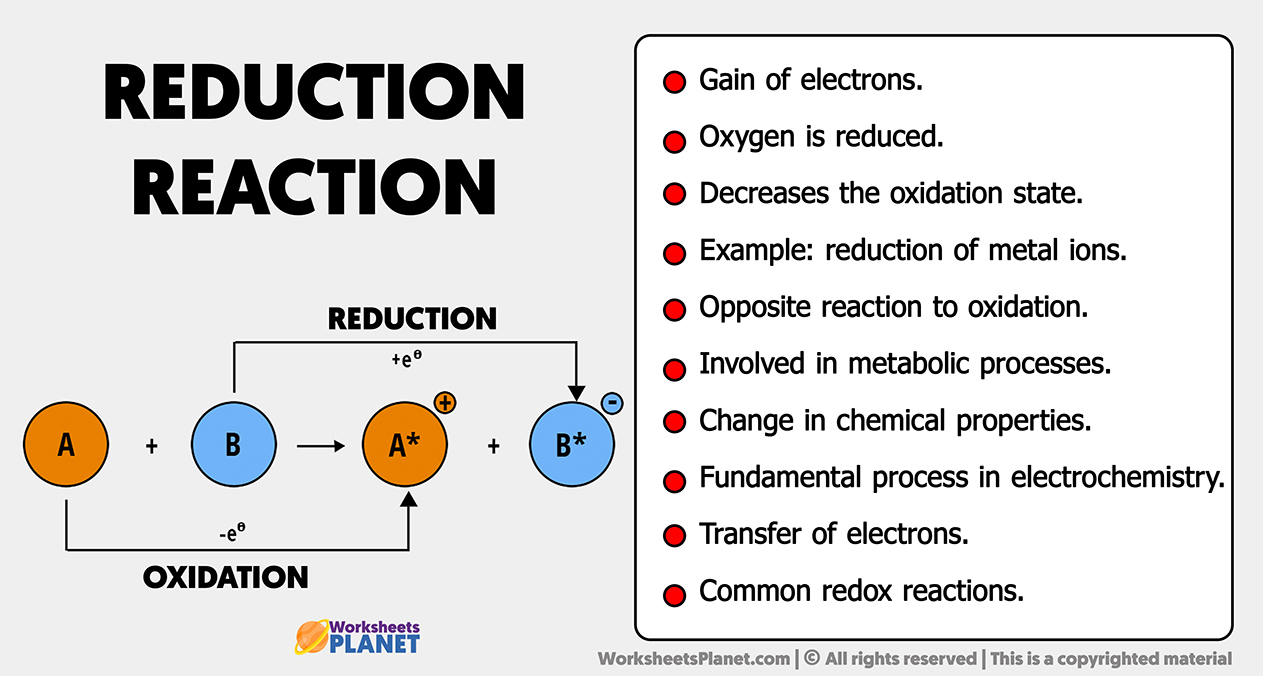
Reduction Reaction Characteristics An oxidation reduction (redox) reaction is a type of chemical reaction that involves a transfer of electrons between two species. an oxidation reduction reaction is any chemical reaction in which the oxidation number of a molecule, atom, or ion changes by gaining or losing an electron. Redox is a type of chemical reaction in which the oxidation states of the reactants change. reduction is the gain of electrons or a decrease in the oxidation state, and occurs simultaneously with oxidation. learn more about redox, electron transfer and atom transfer reactions, and see examples of reduction reactions.

Reduction Oxidation Reaction Redox Reaction Biorender Science Templates What is a redox reaction? a redox reaction is a chemical reaction in which the atoms change their oxidation numbers. some atoms lose electrons and are oxidized – a process known as oxidation. on the other hand, some atoms gain electrons and are reduced – a process known as reduction. An oxidation reduction reaction is any chemical reaction in which the oxidation number of a participating chemical species changes. many such reactions are common and familiar—combustion, rusting, cellular respiration, and photosynthesis are some examples. Reduction is a chemical reaction where a substance gains electrons and decreases its oxidation number. examples of reduction include reactions between zinc and hydrogen ions, and copper oxide with magnesium. This tutorial covers oxidation reduction (redox) reactions in electrochemistry: learn to write simple half reactions and to recognize the oxidizing agent and reducing agent.

Reduction Reaction Reduction is a chemical reaction where a substance gains electrons and decreases its oxidation number. examples of reduction include reactions between zinc and hydrogen ions, and copper oxide with magnesium. This tutorial covers oxidation reduction (redox) reactions in electrochemistry: learn to write simple half reactions and to recognize the oxidizing agent and reducing agent. When the battery is discharging, a chemical reaction occurs at the anode (oxidation) and another at the cathode (reduction). electrons flow from the anode to the cathode through an external circuit, creating an electric current. Learn what a redox reaction is, how to recognize and write it, and the difference between oxidation and reduction. see examples of redox reactions involving metals, nonmetals, oxygen, and other substances. Oxidation and reduction are types of chemical reactions where substances exchange electrons. in oxidation, a substance loses electrons (or gains oxygen), while in reduction, a substance gains electrons (or loses oxygen). Reduction is a process that involves the addition of hydrogen or any other electropositive element or the removal of oxygen or any other electronegative element, according to classical or older concepts.

Reduction Reaction When the battery is discharging, a chemical reaction occurs at the anode (oxidation) and another at the cathode (reduction). electrons flow from the anode to the cathode through an external circuit, creating an electric current. Learn what a redox reaction is, how to recognize and write it, and the difference between oxidation and reduction. see examples of redox reactions involving metals, nonmetals, oxygen, and other substances. Oxidation and reduction are types of chemical reactions where substances exchange electrons. in oxidation, a substance loses electrons (or gains oxygen), while in reduction, a substance gains electrons (or loses oxygen). Reduction is a process that involves the addition of hydrogen or any other electropositive element or the removal of oxygen or any other electronegative element, according to classical or older concepts.
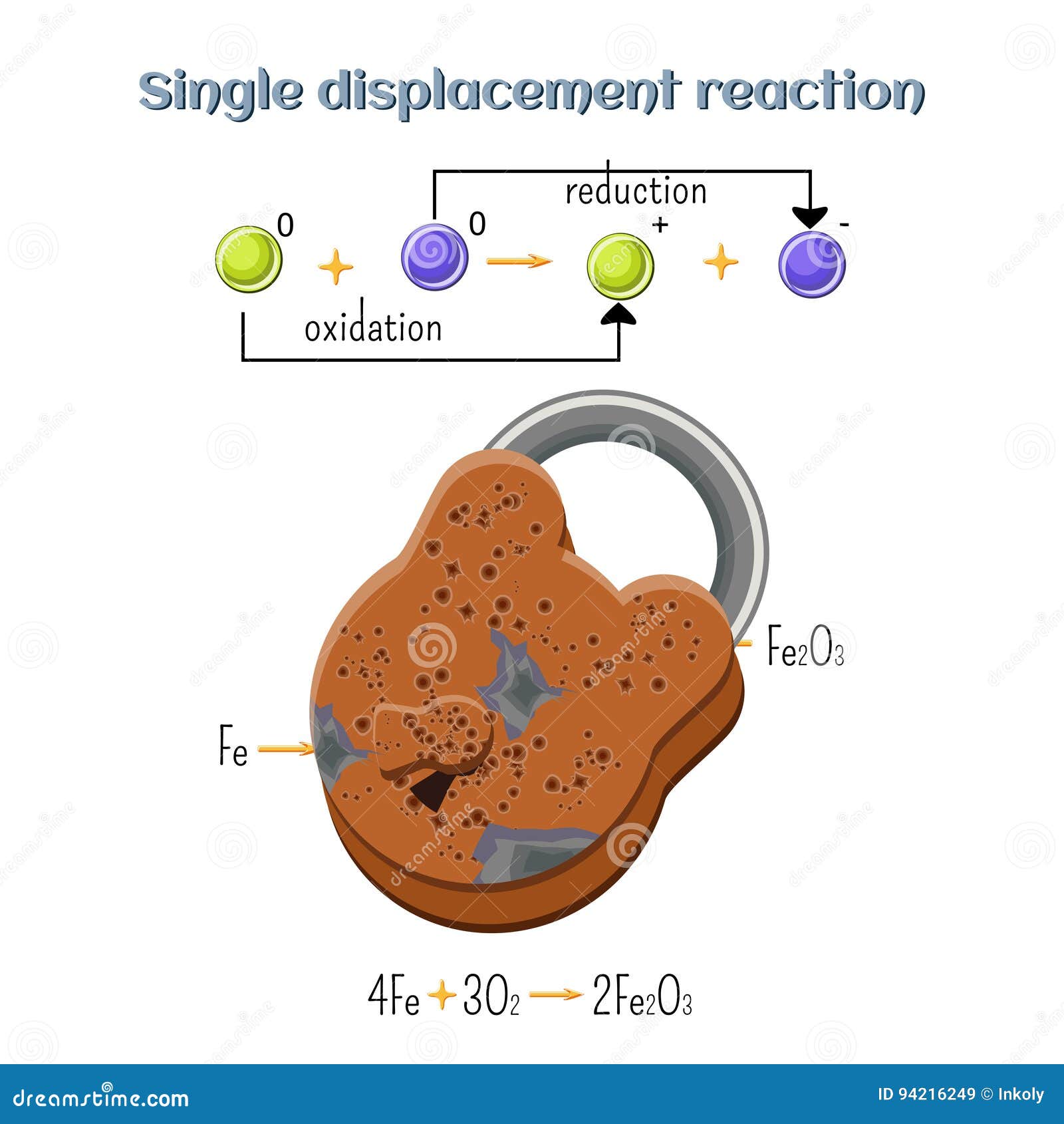
Reduction Reaction Oxidation and reduction are types of chemical reactions where substances exchange electrons. in oxidation, a substance loses electrons (or gains oxygen), while in reduction, a substance gains electrons (or loses oxygen). Reduction is a process that involves the addition of hydrogen or any other electropositive element or the removal of oxygen or any other electronegative element, according to classical or older concepts.
Comments are closed.