Quantum Chemistry 3 7 Particle In A Box Wavefunction Plots

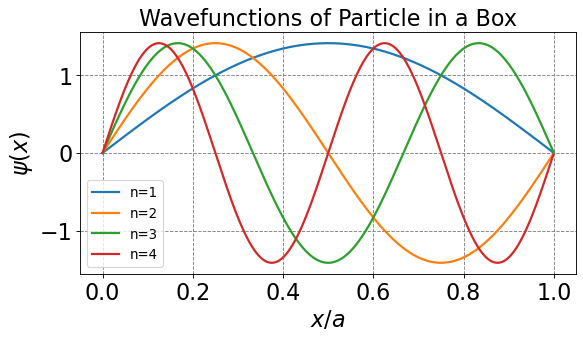
Particle In A Box Quantum Pdf In this section, we apply schrӧdinger’s equation to a particle bound to a one dimensional box. this special case provides lessons for understanding quantum mechanics in more complex systems. the energy of the particle is quantized as a consequence of a standing wave condition inside the box. Short lecture demonstrating particle in a box wavefunctions. the particle in a box is a quantum mechanical model system for a particle which is restricted to a finite region of.

Particle In A 3d Box Pdf Wave Function Hamiltonian Quantum In this section, we apply schrӧdinger’s equation to a particle bound to a one dimensional box. this special case provides lessons for understanding quantum mechanics in more complex systems. the energy of the particle is quantized as a consequence of a standing wave condition inside the box. In this section, we apply schrӧdinger’s equation to a particle bound to a one dimensional box. this special case provides lessons for understanding quantum mechanics in more complex systems. the energy of the particle is quantized as a consequence of a standing wave condition inside the box. Particle in a box is a toy model of electron (or atom, molecule, small quantum object) trapped in some region of space [0, l]. the positional information of a quantum “particle” is described by a quantum wave function ψ (x) which is obtained by solving schrödinger equation with boundary conditions. The particle in the box is a model that can illustrate how a wave equation works. although it does not represent a real situation, we can limit our model to just one dimension (the x dimension, for instance) such that the schrödinger equation becomes significantly simplified.

Particle In A Box Pdf Wave Function Schrödinger Equation Particle in a box is a toy model of electron (or atom, molecule, small quantum object) trapped in some region of space [0, l]. the positional information of a quantum “particle” is described by a quantum wave function ψ (x) which is obtained by solving schrödinger equation with boundary conditions. The particle in the box is a model that can illustrate how a wave equation works. although it does not represent a real situation, we can limit our model to just one dimension (the x dimension, for instance) such that the schrödinger equation becomes significantly simplified. The wave function for a quantum mechanical particle in a box whose walls have arbitrary shape is given by the helmholtz equation subject to the boundary condition that the wave function vanishes at the walls. Cml511 – quantum chemistry 3: particle in a box sameer sapra department of chemistry indian institute of technology delhi. How do we find the wavefunction for the particle in a box or, for that matter, any other system? the wavefunction can be determined by solving the time independent (when the potential is time independent) or time dependent (when the potential is time dependent) schrödinger equation. In this section, we apply schrӧdinger’s equation to a particle bound to a one dimensional box. this special case provides lessons for understanding quantum mechanics in more complex systems. the energy of the particle is quantized as a consequence of a standing wave condition inside the box.

Particle In A Box Problem Quantum Chemistry Pptx The wave function for a quantum mechanical particle in a box whose walls have arbitrary shape is given by the helmholtz equation subject to the boundary condition that the wave function vanishes at the walls. Cml511 – quantum chemistry 3: particle in a box sameer sapra department of chemistry indian institute of technology delhi. How do we find the wavefunction for the particle in a box or, for that matter, any other system? the wavefunction can be determined by solving the time independent (when the potential is time independent) or time dependent (when the potential is time dependent) schrödinger equation. In this section, we apply schrӧdinger’s equation to a particle bound to a one dimensional box. this special case provides lessons for understanding quantum mechanics in more complex systems. the energy of the particle is quantized as a consequence of a standing wave condition inside the box.

Particle In A Box Problem Quantum Chemistry How do we find the wavefunction for the particle in a box or, for that matter, any other system? the wavefunction can be determined by solving the time independent (when the potential is time independent) or time dependent (when the potential is time dependent) schrödinger equation. In this section, we apply schrӧdinger’s equation to a particle bound to a one dimensional box. this special case provides lessons for understanding quantum mechanics in more complex systems. the energy of the particle is quantized as a consequence of a standing wave condition inside the box.
4 5 4 Particle In A Box Chemistry Lessons With Jupyter Notebooks
Comments are closed.