Particle In A Box Pdf Wave Function Schrodinger Equation

Wave Function And Schrodinger Equation Lec1 Pdf Sketching solutions to schrodinger’s equation • estimate the wavefunction for an electron in the 5th energy level of this potential, without solving the schrodinger eq. In the first section of this chapter, we discussed the postulates of quantum mechanics i.e. the step by step procedure to solve a quantum mechanical problem. now it’s the time to implement those rules to the simplest quantum mechanical problem i.e. particle in a one dimensional box.

Spha031 23 A Particle In A Box Infinite Potential Well Ii 2023 The particle in the box model system is the simplest non trivial application of the schrödinger equation, but one which illustrates many of the fundamental concepts of quantum mechanics. Note that the particle is not " xed" localized in space, instead, we can only calculate the "probability" of nding the particle at a position. now let's calculated the averageand "width" spreading of the particle. It details the process of solving the schrödinger equation to obtain the wavefunction and allowed energy values for the particle, highlighting applications in understanding the properties of materials like semiconductors and conjugated organic molecules. We will begin by defining the system and formulate a suitable schrödinger wave equation for it. then we will solve the wave equation to get the eigen functions and subject them to the boundary conditions. we will then normalise the wave functions and analyse them and highlight their significance.

Schrodinger Wave Equation Pdf It details the process of solving the schrödinger equation to obtain the wavefunction and allowed energy values for the particle, highlighting applications in understanding the properties of materials like semiconductors and conjugated organic molecules. We will begin by defining the system and formulate a suitable schrödinger wave equation for it. then we will solve the wave equation to get the eigen functions and subject them to the boundary conditions. we will then normalise the wave functions and analyse them and highlight their significance. Conclusion analytic solution of particle in a box illustrates core quantum concepts. quantized energy, wavefunctions, and expectations obey physical principles. model serves as foundation for understanding more complex systems. Such a system is called a particle in a box. it is one of the most important example quantum systems in chemistry, because it helps us develop intuition about the behavior on electrons confined in molecules. Let us now construct our wave equation by reverse engineering, i.e., we start with a wave function solution and work backwards to obtain the equation. we shall first postulate the wave function for the simplest conceivable system: a free particle. To review, the solutions to the schrödinger equation for the infinite square well and quantum harmonic oscillator were normalizable and labeled by a discrete index n while the solution to the schrödinger equation for the free particle was not normalizable and was labeled by a continuous variable k.

L 15 Solution Of Schrodinger Equation For Particle In A Box Pdf Conclusion analytic solution of particle in a box illustrates core quantum concepts. quantized energy, wavefunctions, and expectations obey physical principles. model serves as foundation for understanding more complex systems. Such a system is called a particle in a box. it is one of the most important example quantum systems in chemistry, because it helps us develop intuition about the behavior on electrons confined in molecules. Let us now construct our wave equation by reverse engineering, i.e., we start with a wave function solution and work backwards to obtain the equation. we shall first postulate the wave function for the simplest conceivable system: a free particle. To review, the solutions to the schrödinger equation for the infinite square well and quantum harmonic oscillator were normalizable and labeled by a discrete index n while the solution to the schrödinger equation for the free particle was not normalizable and was labeled by a continuous variable k.
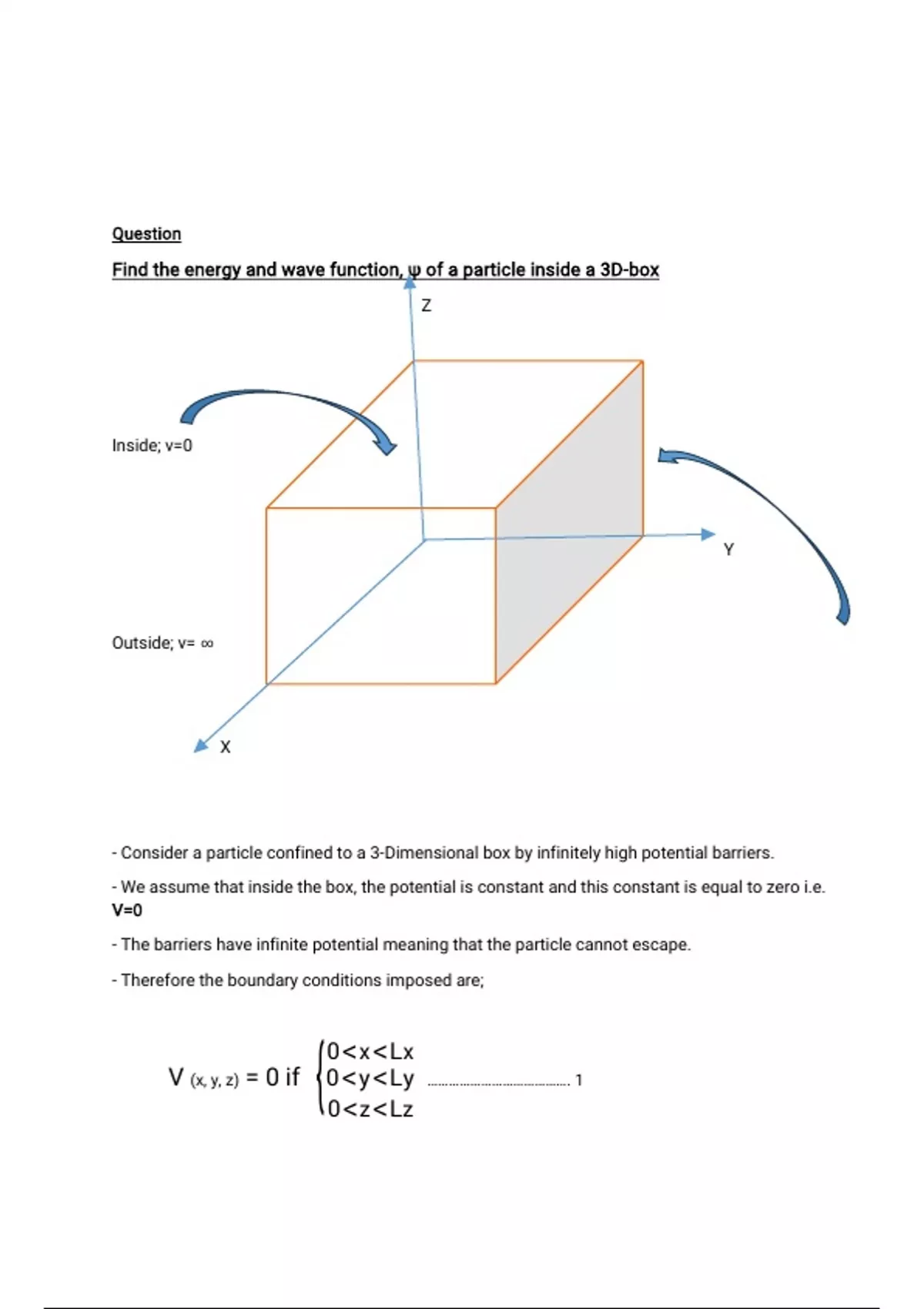
Finding Wave Function And Energy Of A Particle Inside A 3d Box Using Let us now construct our wave equation by reverse engineering, i.e., we start with a wave function solution and work backwards to obtain the equation. we shall first postulate the wave function for the simplest conceivable system: a free particle. To review, the solutions to the schrödinger equation for the infinite square well and quantum harmonic oscillator were normalizable and labeled by a discrete index n while the solution to the schrödinger equation for the free particle was not normalizable and was labeled by a continuous variable k.

Solution Lecture 28 Schrodinger Wave Equation And Its Solution For
Comments are closed.