Particle In A Box Problem Quantum Chemistry

Particle In A Box Quantum Pdf In this chapter, we will develop the theoretical problem of a particle in a box. the purpose here is to explore the capabilities of quantum mechanics and see how some of the mathematical machinery works. Particle in a box is a toy model of electron (or atom, molecule, small quantum object) trapped in some region of space [0, l]. the positional information of a quantum “particle” is described by a quantum wave function ψ (x) which is obtained by solving schrödinger equation with boundary conditions.

5 Particle In Box Pdf The particle in a box model is one of the very few problems in quantum mechanics that can be solved analytically, without approximations. due to its simplicity, the model allows insight into quantum effects without the need for complicated mathematics. Particle in one dimensional box y, v(x) = 0 inside this region.this is called the problem of a free particle in one dimensiona box. this simple model has a crude applic (d2 dx2 )ψ(x) (2me ћ2) ψ(x) = 0 0 ≤ x ≤ a of finding the particle outside the region is zero. Ψ(x) is co. 2. the wave functions and energy levels of the particle are quantized. the wave functions are sinusoidal with n nodes, and the energy is proportional to n^2. 3. the energy levels are spaced further apart at higher n values, with the spacing between levels increasing as the box size decreases. Before we solve that problem we will tackle the simplest of the problems in quantum mechanics, a free particle that does not interact with anything else but it is constained inside a box.

Particle In A Box Problem Quantum Chemistry Pptx 2. the wave functions and energy levels of the particle are quantized. the wave functions are sinusoidal with n nodes, and the energy is proportional to n^2. 3. the energy levels are spaced further apart at higher n values, with the spacing between levels increasing as the box size decreases. Before we solve that problem we will tackle the simplest of the problems in quantum mechanics, a free particle that does not interact with anything else but it is constained inside a box. The particle in a box model is essential for understanding quantum confinement, energy quantization, and the behavior of electrons in nanoscale structures. In this chapter, we solve the time independent schrödinger equation for a very simple system, a particle in a one dimensional box. In this section, we apply schrӧdinger’s equation to a particle bound to a one dimensional box. this special case provides lessons for understanding quantum mechanics in more complex systems. the energy of the particle is quantized as a consequence of a standing wave condition inside the box. Because there is no chance that the particle could ever “escape” an infinite box like this (such an electron would have infinite potential energy!), the wavefunction must be zero outside the box.

Particle In A Box Problem Quantum Chemistry Pptx The particle in a box model is essential for understanding quantum confinement, energy quantization, and the behavior of electrons in nanoscale structures. In this chapter, we solve the time independent schrödinger equation for a very simple system, a particle in a one dimensional box. In this section, we apply schrӧdinger’s equation to a particle bound to a one dimensional box. this special case provides lessons for understanding quantum mechanics in more complex systems. the energy of the particle is quantized as a consequence of a standing wave condition inside the box. Because there is no chance that the particle could ever “escape” an infinite box like this (such an electron would have infinite potential energy!), the wavefunction must be zero outside the box.
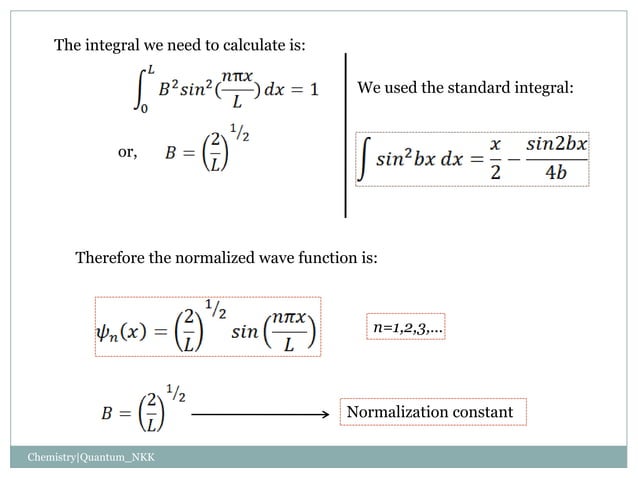
Particle In A Box Problem Quantum Chemistry Pptx In this section, we apply schrӧdinger’s equation to a particle bound to a one dimensional box. this special case provides lessons for understanding quantum mechanics in more complex systems. the energy of the particle is quantized as a consequence of a standing wave condition inside the box. Because there is no chance that the particle could ever “escape” an infinite box like this (such an electron would have infinite potential energy!), the wavefunction must be zero outside the box.
Comments are closed.