Iq Oq Pq What S Needed For Equipment Validation In Life Sciences

Iq Oq Pq What S Needed For Equipment Validation In Life Sciences A comprehensive guide to lab equipment qualification (iq oq pq). learn the process, regulatory standards including fda's finalized csa guidance, and how ai powered automation streamlines validation and compliance. Learn how to effectively manage iq, oq, and pq processes in life sciences equipment validation to ensure compliance and efficiency.

Iq Oq Pq In Equipment Verification And Validation Anab Blog Explore iq, oq, and pq validation in life sciences manufacturing. learn how these steps ensure compliance, improve quality, and enhance patient safety. The sequence of iq, oq, and pq is not arbitrary; it's a logical progression designed to build confidence in equipment performance systematically. each phase relies on the successful completion of the preceding one. Iq, oq, and pq represent three core phases of equipment qualification designed to verify installation accuracy, operational consistency, and performance reliability. Pharmaceutical companies must prioritize quality assurance with the same diligence applied to the installation and utilization of their equipment as they do in the formulation and distribution processes. the gold standard in this domain is the systematic approach defined by iq, oq, and pq.

Iq Oq Pq A Quick Guide To Process Validation 56 Off Iq, oq, and pq represent three core phases of equipment qualification designed to verify installation accuracy, operational consistency, and performance reliability. Pharmaceutical companies must prioritize quality assurance with the same diligence applied to the installation and utilization of their equipment as they do in the formulation and distribution processes. the gold standard in this domain is the systematic approach defined by iq, oq, and pq. Learn the definitions of iq, oq, and pq, key activities, deliverables, and how qms software like simplerqms streamlines equipment qualification in the life sciences. Iq oq pq is one way for laboratories to document objective evidence that equipment or instruments are installed correctly, operate effectively and provide valid results under normal operating conditions. This guide covers equipment qualification requirements, procedures, and best practices to meet fda process validation standards. pharmaceutical companies rely on iq, oq, pq to verify machine performance. Master iq, oq, and pq with this practical guide to equipment validation. learn the differences, see real world examples, and apply proven best practices.
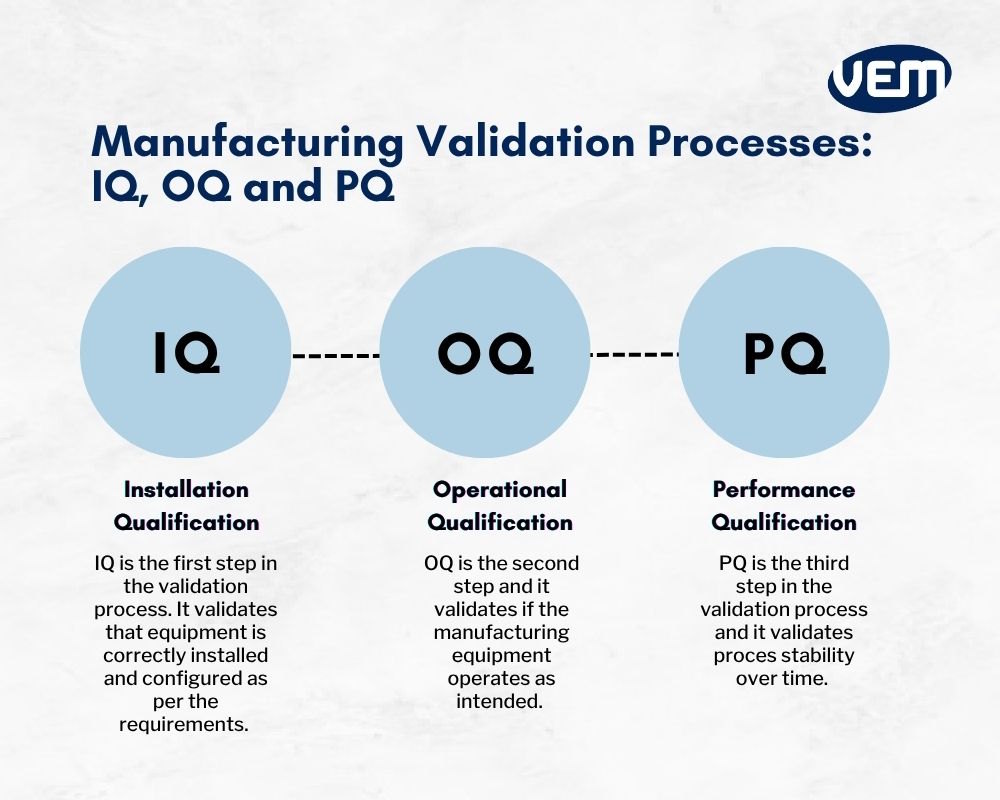
Iq Oq And Pq Validation Learn the definitions of iq, oq, and pq, key activities, deliverables, and how qms software like simplerqms streamlines equipment qualification in the life sciences. Iq oq pq is one way for laboratories to document objective evidence that equipment or instruments are installed correctly, operate effectively and provide valid results under normal operating conditions. This guide covers equipment qualification requirements, procedures, and best practices to meet fda process validation standards. pharmaceutical companies rely on iq, oq, pq to verify machine performance. Master iq, oq, and pq with this practical guide to equipment validation. learn the differences, see real world examples, and apply proven best practices.

Exploring Iq Oq Pq In Pharmaceutical Equipment Validation Blog This guide covers equipment qualification requirements, procedures, and best practices to meet fda process validation standards. pharmaceutical companies rely on iq, oq, pq to verify machine performance. Master iq, oq, and pq with this practical guide to equipment validation. learn the differences, see real world examples, and apply proven best practices.

Exploring Iq Oq Pq In Pharmaceutical Equipment Validation Blog
Comments are closed.