Iq Oq And Pq Validation
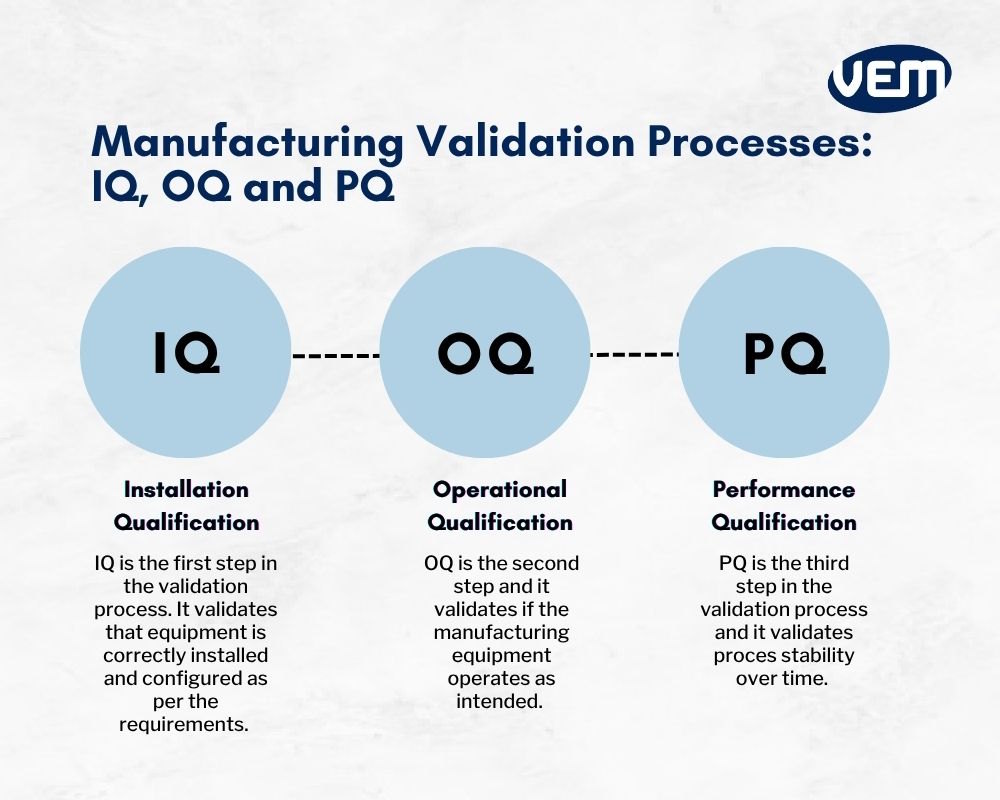
Iq Oq And Pq Validation Get a basic guide to iq, oq, pq for the pharmaceutical, medical device, and diagnostic industries, including fda criteria and a model for resourcing. The common protocols in equipment validation include dq (design qualification), iq (installation qualification), oq (operational qualification), and pq (performance qualification) in pharmaceutical industry.

What Are Iq Oq Pq The 3 Q S Of Software Validation Process Software The dq, iq, oq, pq in pharmaceutical validation process is essential for ensuring product quality, regulatory compliance, and operational efficiency. by following a structured pharmaceutical validation lifecycle, companies can minimize risks, improve productivity, and meet global standards. Iq, oq, and pq are the three essential qualification phases for validating equipment and utilities in regulated life sciences industries — each building on the previous to ensure compliance and product quality. Performance qualification (pq) demonstrates reproducibility and compliance during routine use. together, iq, oq, and pq form a sequential validation framework, with each stage building on the previous one to ensure equipment reliability, regulatory compliance, and product safety. Learn the essentials of iq, oq, and pq for analytical instruments. see real world hplc examples and discover how digital flowmeters like the flowcal 5000 help ensure fda and 21 cfr compliance.
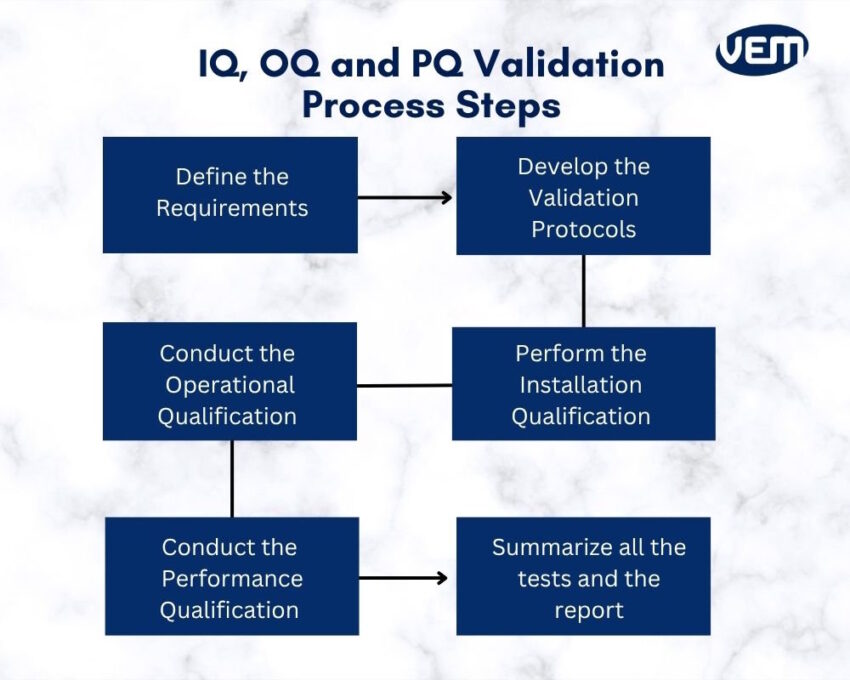
Iq Oq And Pq Validation Performance qualification (pq) demonstrates reproducibility and compliance during routine use. together, iq, oq, and pq form a sequential validation framework, with each stage building on the previous one to ensure equipment reliability, regulatory compliance, and product safety. Learn the essentials of iq, oq, and pq for analytical instruments. see real world hplc examples and discover how digital flowmeters like the flowcal 5000 help ensure fda and 21 cfr compliance. Understand iq, oq, pq phases in pharmaceutical and biotech equipment validation, with compliance focused insight into thermal validation requirements. What is the primary difference between iq, oq, and pq in pharmaceutical labs? iq verifies correct installation, oq confirms the equipment operates within specified ranges, and pq demonstrates consistent performance for its intended routine use over time. Master iq, oq, and pq with this practical guide to equipment validation. learn the differences, see real world examples, and apply proven best practices. Article context: cqv validation dq,iq,oq and pq validation why cqv is essential? cqv implementation cqv challenges in highly regulated industries such as pharmaceuticals and biotechnology, manufacturing is about much more than producing a product. companies must prove through clear documentation and testing that their facilities, systems, and equipment operate correctly and consistently. that.

Fda Part 11 Your Guide To Computer System Validation Csv Compliance Understand iq, oq, pq phases in pharmaceutical and biotech equipment validation, with compliance focused insight into thermal validation requirements. What is the primary difference between iq, oq, and pq in pharmaceutical labs? iq verifies correct installation, oq confirms the equipment operates within specified ranges, and pq demonstrates consistent performance for its intended routine use over time. Master iq, oq, and pq with this practical guide to equipment validation. learn the differences, see real world examples, and apply proven best practices. Article context: cqv validation dq,iq,oq and pq validation why cqv is essential? cqv implementation cqv challenges in highly regulated industries such as pharmaceuticals and biotechnology, manufacturing is about much more than producing a product. companies must prove through clear documentation and testing that their facilities, systems, and equipment operate correctly and consistently. that.
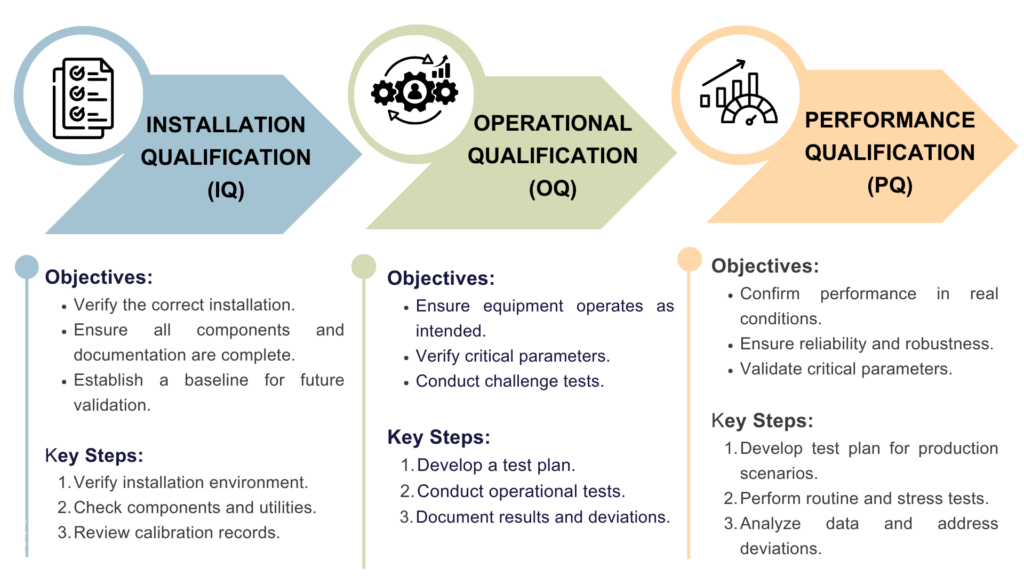
Iq Oq And Pq Importance In Gmp Gmp Insiders Master iq, oq, and pq with this practical guide to equipment validation. learn the differences, see real world examples, and apply proven best practices. Article context: cqv validation dq,iq,oq and pq validation why cqv is essential? cqv implementation cqv challenges in highly regulated industries such as pharmaceuticals and biotechnology, manufacturing is about much more than producing a product. companies must prove through clear documentation and testing that their facilities, systems, and equipment operate correctly and consistently. that.
Comments are closed.