Iq Oq Pq Validation Essential Steps For Life Sciences Manufacturing

Process Validation Iq Oq Pq Pdf Verification And Validation Explore iq, oq, and pq validation in life sciences manufacturing. learn how these steps ensure compliance, improve quality, and enhance patient safety. This guide covers equipment qualification requirements, procedures, and best practices to meet fda process validation standards. pharmaceutical companies rely on iq, oq, pq to verify machine performance.

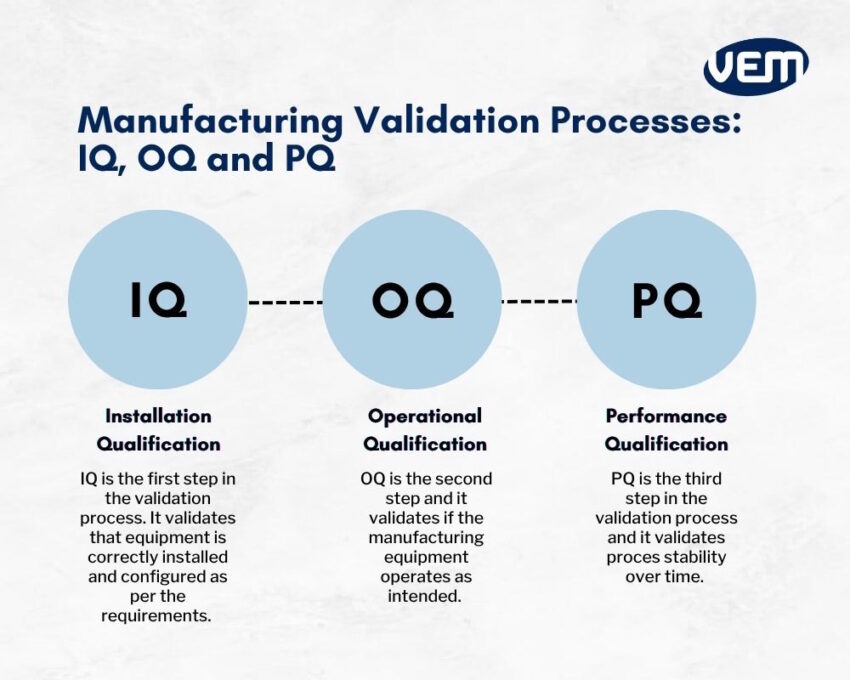
Iq Oq Pq A Quick Guide To Process Validation Pdf Verification Understand iq, oq, pq phases in pharmaceutical and biotech equipment validation, with compliance focused insight into thermal validation requirements. Ensuring equipment quality in pharmaceutical manufacturing involves iq, oq, pq phases. these phases confirm correct installation, consistent performance, and meeting user needs. What is the primary difference between iq, oq, and pq in pharmaceutical labs? iq verifies correct installation, oq confirms the equipment operates within specified ranges, and pq demonstrates consistent performance for its intended routine use over time. Together, iq, oq, and pq form a sequential validation framework, with each stage building on the previous one to ensure equipment reliability, regulatory compliance, and product safety.

Guide To Iq Oq Pq For The Pharmaceutical Industry Pdf What is the primary difference between iq, oq, and pq in pharmaceutical labs? iq verifies correct installation, oq confirms the equipment operates within specified ranges, and pq demonstrates consistent performance for its intended routine use over time. Together, iq, oq, and pq form a sequential validation framework, with each stage building on the previous one to ensure equipment reliability, regulatory compliance, and product safety. The validation steps—installation qualification (iq), operational qualification (oq), and performance qualification (pq)—are vital for confirming that equipment and systems are installed and functioning correctly and consistently. Iq, oq, pq are vital qualification steps in pharma production. learn how they ensure equipment compliance, product quality, and gmp regulatory success. These are the abbreviations we use in the medical device industry for the three steps of process validation: installation qualification (iq), operational qualification (oq), and performance qualification (pq). These three qualification protocols, iq, oq, and pq, are commonly employed in the health and life sciences industries to ensure the quality, reliability, and compliance of equipment,.

Iq Oq Pq Validation Essential Steps For Life Sciences Manufacturing The validation steps—installation qualification (iq), operational qualification (oq), and performance qualification (pq)—are vital for confirming that equipment and systems are installed and functioning correctly and consistently. Iq, oq, pq are vital qualification steps in pharma production. learn how they ensure equipment compliance, product quality, and gmp regulatory success. These are the abbreviations we use in the medical device industry for the three steps of process validation: installation qualification (iq), operational qualification (oq), and performance qualification (pq). These three qualification protocols, iq, oq, and pq, are commonly employed in the health and life sciences industries to ensure the quality, reliability, and compliance of equipment,.

Iq Oq Pq Validation Essential Steps For Life Sciences Manufacturing These are the abbreviations we use in the medical device industry for the three steps of process validation: installation qualification (iq), operational qualification (oq), and performance qualification (pq). These three qualification protocols, iq, oq, and pq, are commonly employed in the health and life sciences industries to ensure the quality, reliability, and compliance of equipment,.
Iq Oq And Pq Validation
Comments are closed.