How Does Evaporation Cause Cooling Geeksforgeeks
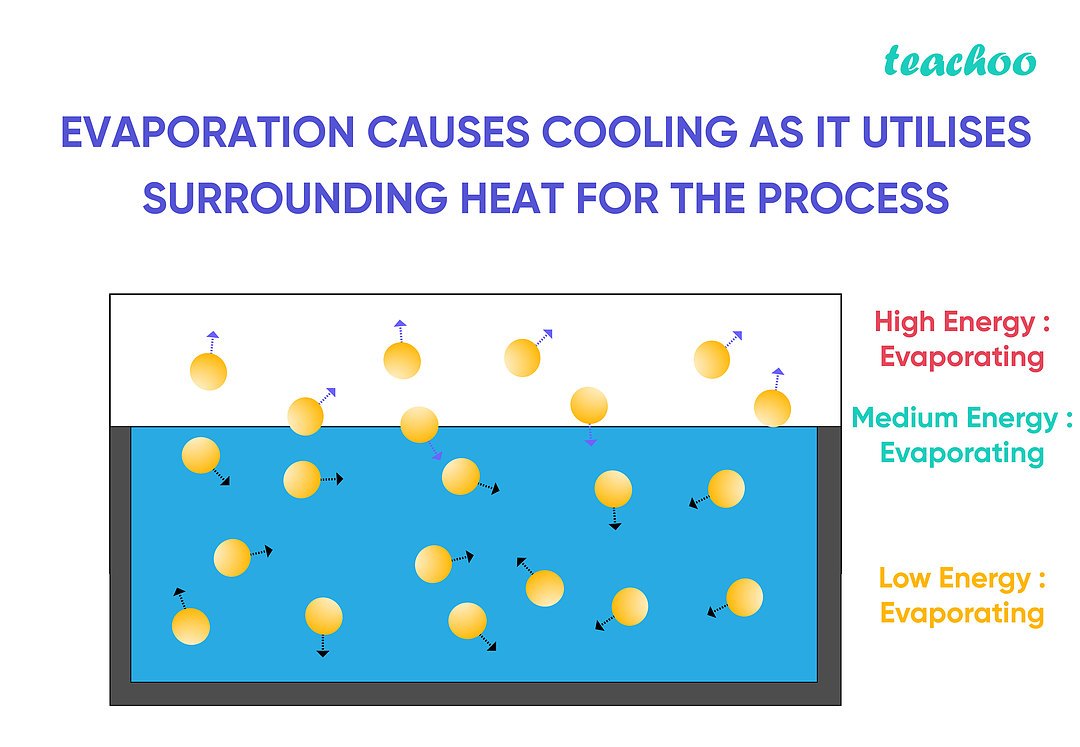
How Does Evaporation Cause Cooling Explain With Examples Teachoo But in modern times we know that evaporation cause cooling because the particles (atoms and molecules) present at the surface of the liquid surface absorb energy from their surroundings and transform it into vapour, which then causes the cooling effect. Because the higher energy molecules escape, the remaining molecules have lower average energy, which results in a drop in temperature and produces a cooling effect.

What Is Evaporation Definition Facts Process Examples With Videos Evaporation** ** causes cooling because the process requires heat energy. the energy is taken away by the molecules when they convert from liquid into gas, and this causes cooling on the original surface. During evaporation, the. Discover how evaporation causes cooling with simple explanations, real life examples, and student friendly science concepts. learn why surfaces feel cooler as liquid turns to vapor. The key principle for evaporative cooling is that the phase change from a liquid to a gas absorbs energy from the surroundings. it works for all liquids changing into vapor, but water is especially effective because of its high heat of vaporization.
Evaporation Causes Cooling Definition Explanation 52 Off Discover how evaporation causes cooling with simple explanations, real life examples, and student friendly science concepts. learn why surfaces feel cooler as liquid turns to vapor. The key principle for evaporative cooling is that the phase change from a liquid to a gas absorbs energy from the surroundings. it works for all liquids changing into vapor, but water is especially effective because of its high heat of vaporization. Evaporation causes cooling because the fastest moving, high energy molecules leave the liquid and take heat away with them. this reduces the average energy of the remaining molecules, lowering the temperature of the liquid and its surroundings. On a hot day, the water molecules in your perspiration absorb body heat and evaporate from the surface of your skin. the evaporation process leaves the remaining perspiration cooler, which in turn absorbs more heat from your body. a given liquid will evaporate more quickly when it is heated. Evaporation causes cooling because the particles of a liquid absorb heat energy from their surroundings to change into vapour. during evaporation, the higher energy (faster moving) molecules escape from the surface of the liquid, leaving behind molecules with lower average kinetic energy. This process involves the evaporation of a liquid, most commonly water, which absorbs heat from its surroundings and creates a cooling effect. this principle is important in biological systems and industrial applications.

Evaporation Causes Cooling Definition Explanation 52 Off Evaporation causes cooling because the fastest moving, high energy molecules leave the liquid and take heat away with them. this reduces the average energy of the remaining molecules, lowering the temperature of the liquid and its surroundings. On a hot day, the water molecules in your perspiration absorb body heat and evaporate from the surface of your skin. the evaporation process leaves the remaining perspiration cooler, which in turn absorbs more heat from your body. a given liquid will evaporate more quickly when it is heated. Evaporation causes cooling because the particles of a liquid absorb heat energy from their surroundings to change into vapour. during evaporation, the higher energy (faster moving) molecules escape from the surface of the liquid, leaving behind molecules with lower average kinetic energy. This process involves the evaporation of a liquid, most commonly water, which absorbs heat from its surroundings and creates a cooling effect. this principle is important in biological systems and industrial applications.

Evaporation Causes Cooling Definition Explanation 52 Off Evaporation causes cooling because the particles of a liquid absorb heat energy from their surroundings to change into vapour. during evaporation, the higher energy (faster moving) molecules escape from the surface of the liquid, leaving behind molecules with lower average kinetic energy. This process involves the evaporation of a liquid, most commonly water, which absorbs heat from its surroundings and creates a cooling effect. this principle is important in biological systems and industrial applications.

How Does Evaporation Cause Cooling The Underlying Principle Behind
Comments are closed.