How Does Evaporation Really Work

How Does Evaporation Molecular Work At Rafaela Woodruff Blog When water is heated, it e vaporates. the molecules move and vibrate so quickly that they escape into the atmosphere as molecules of water vapor. e vaporation is a very important part of the water cycle. heat from the sun, or solar energy, powers the e vaporation process. What is evaporation and why does it occur? evaporation is the process that changes liquid water to gaseous water (water vapor). water moves from the earth’s surface to the atmosphere via evaporation. evaporation occurs when energy (heat) forces the bonds that hold water molecules together to break.
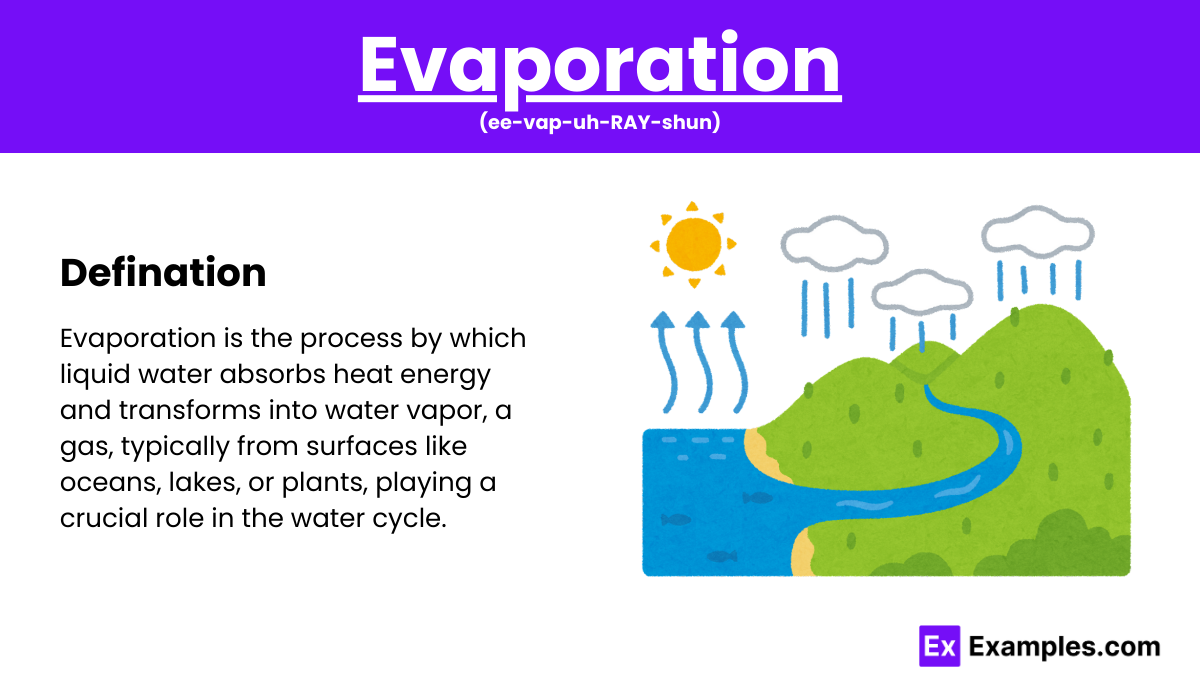
Evaporation Examples Otemoto Evaporation, mostly from the oceans and from vegetation, replenishes the humidity of the air. it is an important part of the exchange of energy in the earth atmosphere system that produces atmospheric motion and therefore weather and climate. Researchers at mit and in tokyo reveal fundamental characteristics of how evaporation works, which could help in the design of power plants and cooling systems. The evaporation process leaves the remaining perspiration cooler, which in turn absorbs more heat from your body. a given liquid will evaporate more quickly when it is heated. Water does not truly disappear when it dries; instead, it changes its physical state from a liquid to an invisible gas known as water vapor. this transformation is called evaporation, and it occurs at the molecular level. water molecules are in constant, random motion, possessing kinetic energy.

Evaporation Examples Otemoto The evaporation process leaves the remaining perspiration cooler, which in turn absorbs more heat from your body. a given liquid will evaporate more quickly when it is heated. Water does not truly disappear when it dries; instead, it changes its physical state from a liquid to an invisible gas known as water vapor. this transformation is called evaporation, and it occurs at the molecular level. water molecules are in constant, random motion, possessing kinetic energy. What is evaporation? to begin with, evaporation can be defined as the process by which a liquid changes into vapor at temperatures below its boiling point. this transformation occurs at the surface of the liquid and does not require the entire substance to heat up to its boiling temperature. For the first time, mit scientists have analyzed the evaporation process in detail at a molecular level and determined the physics of evaporation. And what simple thing can we do to take action about global warming? the chemistry and science of evaporation is explained in this video with 3d animations. Evaporation is the process where a liquid changes to a gas. it happens when single particles at the surface of a liquid have enough energy to break away from the other particles.
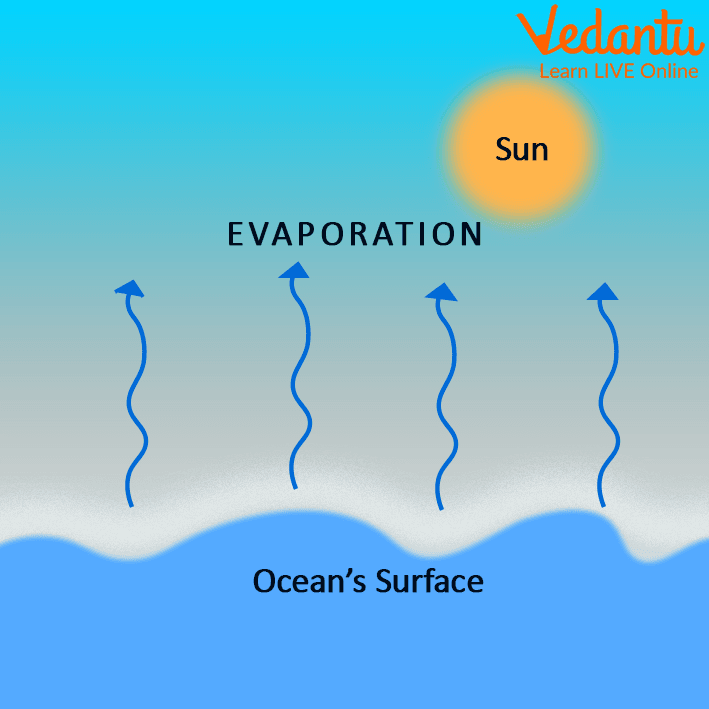
Evaporation Examples Otemoto What is evaporation? to begin with, evaporation can be defined as the process by which a liquid changes into vapor at temperatures below its boiling point. this transformation occurs at the surface of the liquid and does not require the entire substance to heat up to its boiling temperature. For the first time, mit scientists have analyzed the evaporation process in detail at a molecular level and determined the physics of evaporation. And what simple thing can we do to take action about global warming? the chemistry and science of evaporation is explained in this video with 3d animations. Evaporation is the process where a liquid changes to a gas. it happens when single particles at the surface of a liquid have enough energy to break away from the other particles.
Comments are closed.