Evaporation Causes Cooling

What Is Evaporation Definition Facts Process Examples With Videos Discover how evaporation causes cooling with simple explanations, real life examples, and student friendly science concepts. learn why surfaces feel cooler as liquid turns to vapor. But in modern times we know that evaporation cause cooling because the particles (atoms and molecules) present at the surface of the liquid surface absorb energy from their surroundings and transform it into vapour, which then causes the cooling effect.
Evaporation Causes Cooling Definition Explanation 52 Off Evaporation** ** causes cooling because the process requires heat energy. the energy is taken away by the molecules when they convert from liquid into gas, and this causes cooling on the original surface. During evaporation, liquid molecules absorb energy in the form of heat. this energy absorption leads to a cooling effect because the molecules with higher kinetic energy leave the liquid, reducing the average kinetic energy of the remaining molecules. But why does the evaporation of sweat cool you down? when a liquid such as sweat evaporates, energetic particles on the surface of the liquid escape into the air. Evaporation is an endothermic process, meaning it requires energy input. the energy needed for particles to break free from the liquid comes from the surrounding environment — usually the remaining liquid or the surface it touches. this removal of energy contributes further to the cooling effect.
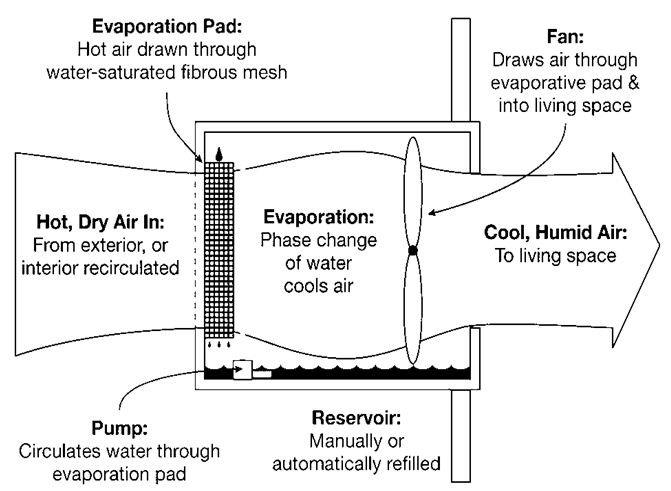
Evaporation Causes Cooling Process And Applications Of Evaporation But why does the evaporation of sweat cool you down? when a liquid such as sweat evaporates, energetic particles on the surface of the liquid escape into the air. Evaporation is an endothermic process, meaning it requires energy input. the energy needed for particles to break free from the liquid comes from the surrounding environment — usually the remaining liquid or the surface it touches. this removal of energy contributes further to the cooling effect. Evaporation causes cooling because the particles of a liquid absorb heat energy from their surroundings to change into vapour. during evaporation, the higher energy (faster moving) molecules escape from the surface of the liquid, leaving behind molecules with lower average kinetic energy. The energy absorbed from the vaporised liquid as it evaporates causes the temperature of the liquid to drop, resulting in evaporative cooling. let us read the article to learn more about the evaporation process with examples. Evaporation causes cooling because the fastest moving, high energy molecules leave the liquid and take heat away with them. this reduces the average energy of the remaining molecules, lowering the temperature of the liquid and its surroundings. Evaporation is a cooling process because when a liquid turns to a gas, it requires more energy, which it must obtain from its surroundings. the energy is in the form of heat, and as the heat energy evaporates, the surroundings cool.

How Does Evaporation Cause Cooling Geeksforgeeks Evaporation causes cooling because the particles of a liquid absorb heat energy from their surroundings to change into vapour. during evaporation, the higher energy (faster moving) molecules escape from the surface of the liquid, leaving behind molecules with lower average kinetic energy. The energy absorbed from the vaporised liquid as it evaporates causes the temperature of the liquid to drop, resulting in evaporative cooling. let us read the article to learn more about the evaporation process with examples. Evaporation causes cooling because the fastest moving, high energy molecules leave the liquid and take heat away with them. this reduces the average energy of the remaining molecules, lowering the temperature of the liquid and its surroundings. Evaporation is a cooling process because when a liquid turns to a gas, it requires more energy, which it must obtain from its surroundings. the energy is in the form of heat, and as the heat energy evaporates, the surroundings cool.

Evaporation Causes Cooling Experiment At Eusebio Gonzalez Blog Evaporation causes cooling because the fastest moving, high energy molecules leave the liquid and take heat away with them. this reduces the average energy of the remaining molecules, lowering the temperature of the liquid and its surroundings. Evaporation is a cooling process because when a liquid turns to a gas, it requires more energy, which it must obtain from its surroundings. the energy is in the form of heat, and as the heat energy evaporates, the surroundings cool.
Comments are closed.