Double Displacement Sodium Sulfite And Hcl
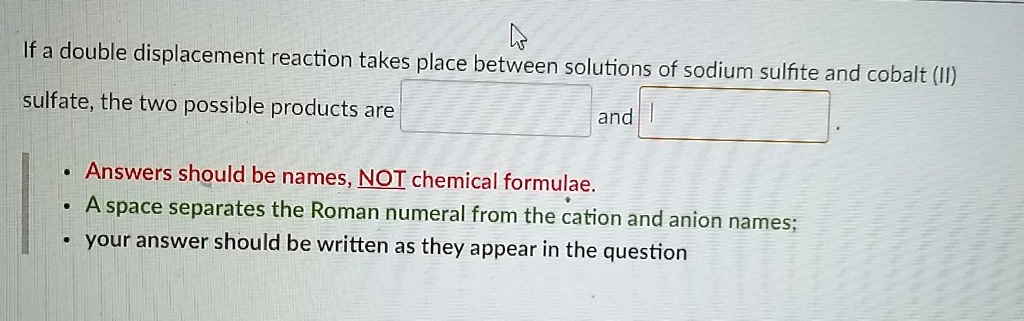
Solved If A Double Displacement Reaction Takes Place Between Solutions Part of ncssm core collection: this video shows the double displacement reaction of na2so3 and hcl. dlt.ncssm.edu more. Na2so3 hcl = nacl h2so3 is a double displacement (metathesis) reaction where one mole of aqueous sodium sulfite [na 2 so 3] and two moles of aqueous hydrogen chloride [hcl] react to form two moles of aqueous sodium chloride [nacl] and one mole of aqueous sulfurous acid [h 2 so 3].

Sodium Sulfite Sodium Sulphite Latest Price Manufacturers Suppliers Double displacement reactions generally occur between substances in an aqueous solution. in order for a reaction to occur, one of the products is usually a solid precipitate, a gas, or a molecular compound such as water. watch this video for an explanation of double displacement reactions. Some double replacement reactions produce a gaseous product which then bubbles out of the solution and escapes into the air. when solutions of sodium sulfide and hydrochloric acid are mixed, the products of the reaction are aqueous sodium chloride and hydrogen sulfide gas. Hence, we see the reaction between dil. hcl and sodium sulphite is a type of double displacement reaction which will lead to formation of sodium chloride, water and sulphur dioxide gas. note: sulphur dioxide gas is highly soluble in water and forms a strong acid known as sulphuric acid. For each element, we check if the number of atoms is balanced on both sides of the equation. na is not balanced: 2 atoms in reagents and 1 atom in products. s is balanced: 1 atom in reagents and 1 atom in products. o is balanced: 3 atoms in reagents and 3 atoms in products. h is not balanced: 1 atom in reagents and 2 atoms in products.
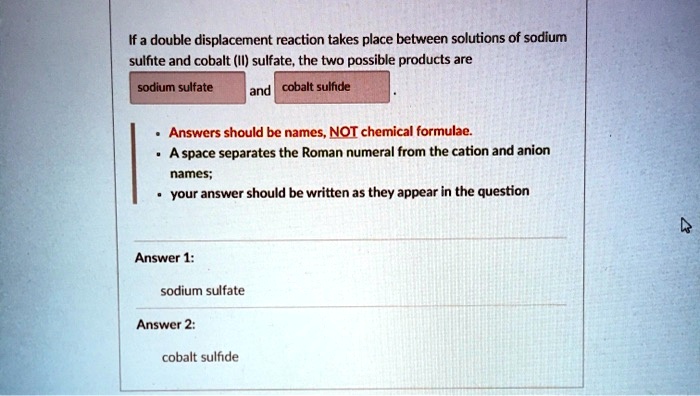
If A Double Displacement Reaction Takes Place Between Solutions Of Hence, we see the reaction between dil. hcl and sodium sulphite is a type of double displacement reaction which will lead to formation of sodium chloride, water and sulphur dioxide gas. note: sulphur dioxide gas is highly soluble in water and forms a strong acid known as sulphuric acid. For each element, we check if the number of atoms is balanced on both sides of the equation. na is not balanced: 2 atoms in reagents and 1 atom in products. s is balanced: 1 atom in reagents and 1 atom in products. o is balanced: 3 atoms in reagents and 3 atoms in products. h is not balanced: 1 atom in reagents and 2 atoms in products. Solved and balanced chemical equation na2so3 2 hcl → h2o 2 nacl so2 with completed products. application for completing products and balancing equations. Introduction: you will study double displacement reactions using a small scale method and predict the products of double displacement reactions. background: you will combine two water solutions, each containing positive and negative ions. ### step 3: analyze the reaction in this reaction, sodium (na) is more reactive than hydrogen (h), so it displaces hydrogen from hydrochloric acid, resulting in the formation of sodium chloride (nacl). When sodium sulfide (na 2 s) and hydrochloric acid (hcl) solutions are combined, the reaction produces aqueous sodium chloride (nacl) and hydrogen sulfide (h 2 s) gas.

Double Displacement Reaction Of Ammonium Chloride And Sodium Hydroxide Solved and balanced chemical equation na2so3 2 hcl → h2o 2 nacl so2 with completed products. application for completing products and balancing equations. Introduction: you will study double displacement reactions using a small scale method and predict the products of double displacement reactions. background: you will combine two water solutions, each containing positive and negative ions. ### step 3: analyze the reaction in this reaction, sodium (na) is more reactive than hydrogen (h), so it displaces hydrogen from hydrochloric acid, resulting in the formation of sodium chloride (nacl). When sodium sulfide (na 2 s) and hydrochloric acid (hcl) solutions are combined, the reaction produces aqueous sodium chloride (nacl) and hydrogen sulfide (h 2 s) gas.
In This Double Displacement Reaction Two Compounds Sodium Hydroxide And ### step 3: analyze the reaction in this reaction, sodium (na) is more reactive than hydrogen (h), so it displaces hydrogen from hydrochloric acid, resulting in the formation of sodium chloride (nacl). When sodium sulfide (na 2 s) and hydrochloric acid (hcl) solutions are combined, the reaction produces aqueous sodium chloride (nacl) and hydrogen sulfide (h 2 s) gas.
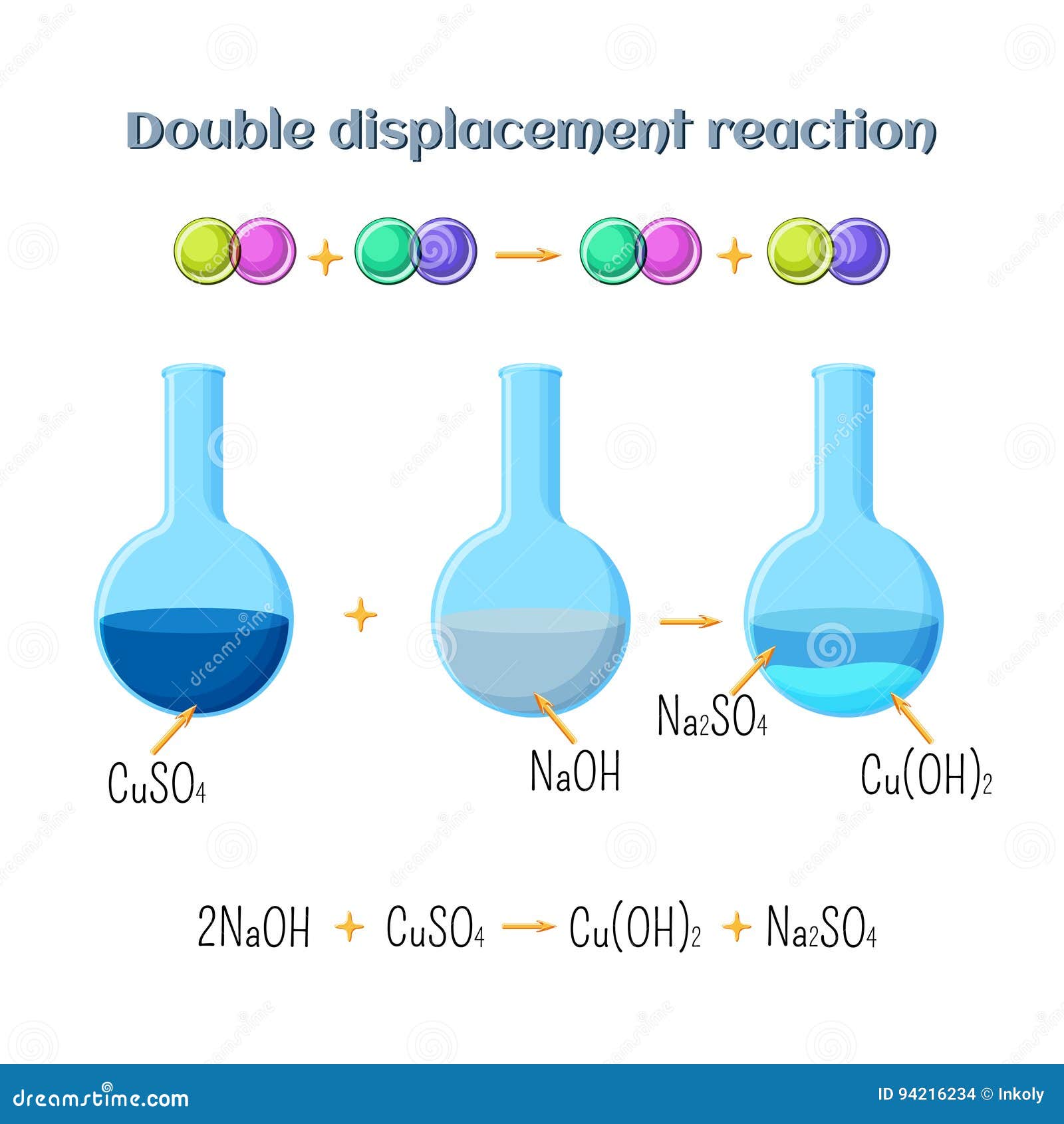
Double Displacement Reaction Sodium Hydroxide Copper Sulfate Types
Comments are closed.