Double Displacement Reaction Definition Examples Types Teachoo
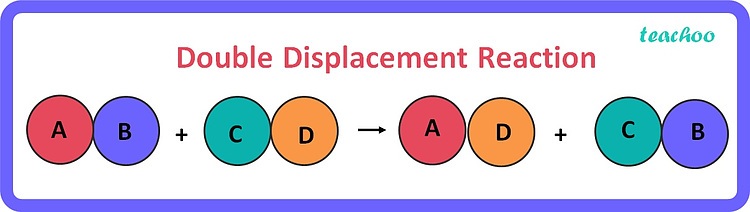
Double Displacement Reaction Definition Examples Types Teachoo It is a type of chemical reaction. A double displacement reaction is a type of chemical reaction in which two reactants exchange ions to generate two new molecules. the double displacement reaction occurs in an aqueous solution and usually leads in the formation of a precipitate.

Double Displacement Reaction Definition Examples Types Teachoo What is displacement reaction?. Learn displacement reactions in chemistry with clear definitions, single vs double types, equations, and real life examples for cbse exams. Learn about double displacement reactions (often called salt metathesis) in chemistry and see examples of representative chemical reactions. There are three types of double displacement reactions: formation of a precipitate, formation of gas, and formation of salt and water.
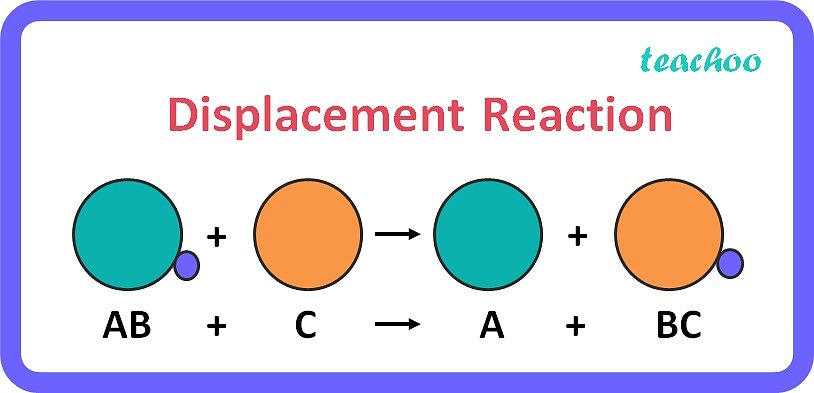
Displacement Reaction Definition Types Reactivity Series Teachoo Learn about double displacement reactions (often called salt metathesis) in chemistry and see examples of representative chemical reactions. There are three types of double displacement reactions: formation of a precipitate, formation of gas, and formation of salt and water. Learn about double replacement reaction definition, or as also known as double displacement reaction. read about different types, how to do double replacement reactions, and. What is the difference between. There are 5 types of chemical. A double displacement reaction involves two ionic compounds in water where the ions switch places to form new ionic compounds. there are three types of chemical equations that can be written: 1) overall, 2) complete ionic, and 3) net ionic.

Double Replacement Reaction Definition Types Examples Lesson Learn about double replacement reaction definition, or as also known as double displacement reaction. read about different types, how to do double replacement reactions, and. What is the difference between. There are 5 types of chemical. A double displacement reaction involves two ionic compounds in water where the ions switch places to form new ionic compounds. there are three types of chemical equations that can be written: 1) overall, 2) complete ionic, and 3) net ionic.
Comments are closed.