Double Displacement Reaction Sodium Hydroxide Copper Sulfate Types

Double Displacement Reaction Sodium Hydroxide And Copper Sulfate The reaction between copper (ii) sulfate (cuso₄) and sodium hydroxide (naoh) is classified as a double displacement reaction, also known as a double replacement or metathesis reaction. Cuso4 naoh = cu (oh)2 na2so4 is a double displacement (metathesis) reaction where one mole of aqueous cupric sulfate [cuso 4] and two moles of aqueous sodium hydroxide [naoh] react to form one mole of solid cupric hydroxide [cu (oh) 2] and one mole of aqueous sodium sulfate [na 2 so 4].
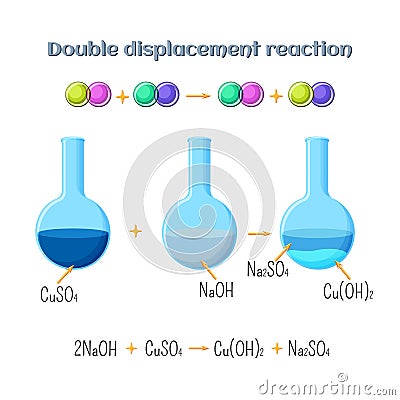
Double Displacement Reaction Sodium Hydroxide And Copper Sulfate Predict the products for the following single and double displacement reactions, and write balanced molecular equations (including physical states) for each of them. The experiment results present five stages of the copper reaction cycle, stating chemical reactions and equations, and types of reactions, starting with copper rods dissolving in nitric. This reaction is a double displacement reaction where copper (ii) sulfate ( (\ce {cuso4})) reacts with sodium hydroxide ( (\ce {naoh})) to form copper (ii) hydroxide ( (\ce {cu (oh)2})), a solid precipitate, and sodium sulfate ( (\ce {na2so4})), which remains in solution. There are three types of double displacement reactions: formation of a precipitate, formation of gas, and formation of salt and water.

Double Displacement Reaction Sodium Hydroxide And Copper Sulfate This reaction is a double displacement reaction where copper (ii) sulfate ( (\ce {cuso4})) reacts with sodium hydroxide ( (\ce {naoh})) to form copper (ii) hydroxide ( (\ce {cu (oh)2})), a solid precipitate, and sodium sulfate ( (\ce {na2so4})), which remains in solution. There are three types of double displacement reactions: formation of a precipitate, formation of gas, and formation of salt and water. Copper (n) sulfate, cuso4, reacts with sodium hydroxide, naoh, in a double displacement reaction. a precipitate of copper (n) hydroxide, cu (oh)2, and aqueous sodium sulfate, na2s04, is produced. Sodium hydroxide with copper (ii) sulfate here, copper (ii) sulfate (cuso 4) is added to sodium hydroxide (naoh). the result is a blue precipitate. (duration 7 seconds, size 401 k, file name movies naoh naohcuso.mov). Keith ramsey and rick crosslin demonstrate the double displacement reaction of copper sulfate & sodium hydroxide. cuso4 naoh→cu (oh)2 na2so4. Copper (ii) sulfate reacts with sodium hydroxide in a double displacement reaction. the equation for the reaction is : cuso4 (a q). 2naoh (aq) >cu (oh)2 (aq). na2so4 (aq).50.0ml of 0.300 m di l copper (ii) sulfate is mixed with an equal volume of 0.600 mol l sodium hydroxide.
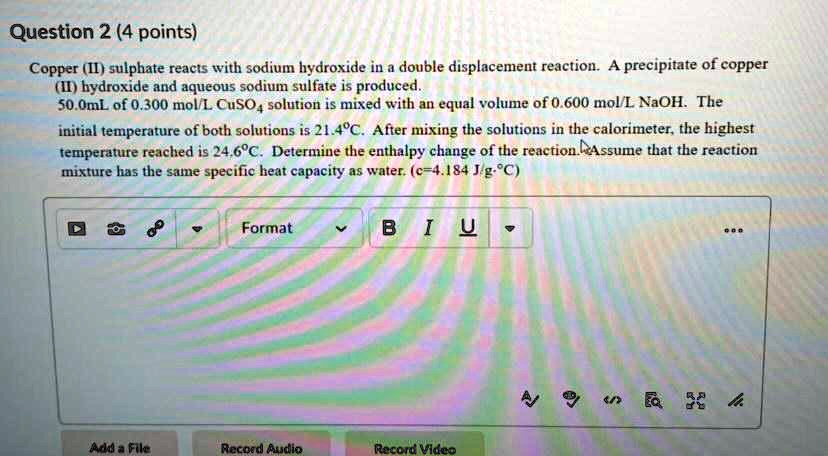
Solved Question 2 4 Points Copper Ii Sulphate Reacts With Sodium Copper (n) sulfate, cuso4, reacts with sodium hydroxide, naoh, in a double displacement reaction. a precipitate of copper (n) hydroxide, cu (oh)2, and aqueous sodium sulfate, na2s04, is produced. Sodium hydroxide with copper (ii) sulfate here, copper (ii) sulfate (cuso 4) is added to sodium hydroxide (naoh). the result is a blue precipitate. (duration 7 seconds, size 401 k, file name movies naoh naohcuso.mov). Keith ramsey and rick crosslin demonstrate the double displacement reaction of copper sulfate & sodium hydroxide. cuso4 naoh→cu (oh)2 na2so4. Copper (ii) sulfate reacts with sodium hydroxide in a double displacement reaction. the equation for the reaction is : cuso4 (a q). 2naoh (aq) >cu (oh)2 (aq). na2so4 (aq).50.0ml of 0.300 m di l copper (ii) sulfate is mixed with an equal volume of 0.600 mol l sodium hydroxide.
Comments are closed.