Atomic Size Ions Atomic Size Electronegativity

Atomic Size Ions Atomic Size Electronegativity As a result, atoms and ions cannot be said to have exact sizes; however, some atoms are larger or smaller than others, and this influences their chemistry. in this section, we discuss how atomic and ion “sizes” are defined and obtained. Learn about periodic table trends: atomic size, ionization energy, and electronegativity. understand how they change across periods and groups.
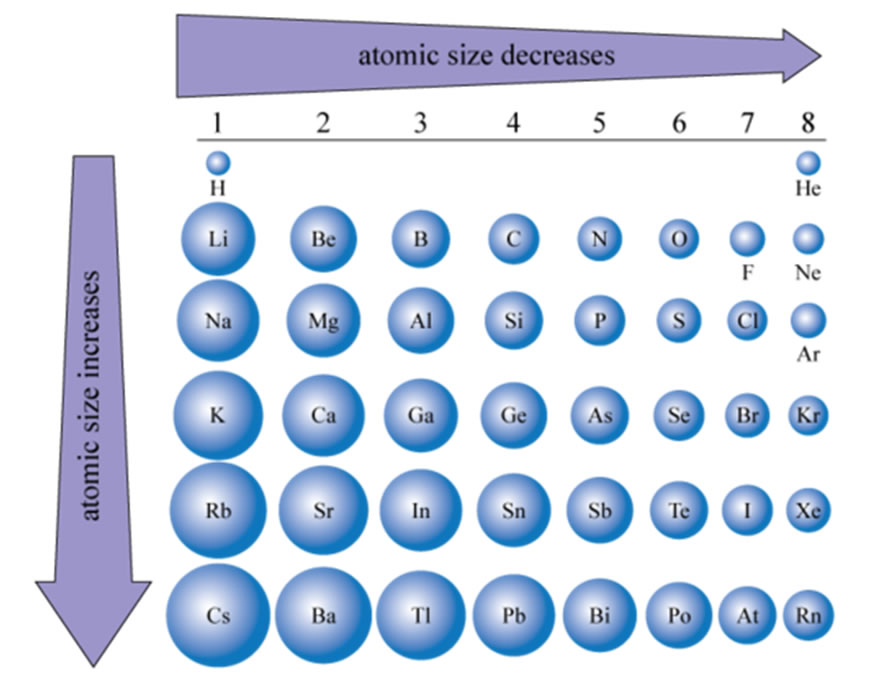
Dimensions Of Atoms And Ions How To Determine Atomic Size Ummlr Comprehensive revision notes for atomic ionic size, ie, ea, electronegativity in periodic law & trends for chemistry in waec waec nigeria. master this topic with our detailed study materials and get personalized help from our ai tutor to answer questions and mark your work. With just a few clicks, you can create three dimensional versions of the periodic table showing atomic size or graphs of ionization energies from all measured elements. Certain properties—notably atomic radius, ionization energies, and electron affinities—can be qualitatively understood by the positions of the elements on the periodic table. Okay, let's break down the relationship between atomic size, ionization energy, and electronegativity. these three properties are fundamentally linked and understanding their interplay is crucial in chemistry.

Electronegativity Ions Atomic Size Electronegativity Certain properties—notably atomic radius, ionization energies, and electron affinities—can be qualitatively understood by the positions of the elements on the periodic table. Okay, let's break down the relationship between atomic size, ionization energy, and electronegativity. these three properties are fundamentally linked and understanding their interplay is crucial in chemistry. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. the higher the associated electronegativity, the more an atom or a substituent group attracts electrons. Understanding the trends in group properties such as atomic size, ionization energy, electronegativity, and electron affinity is fundamental to unraveling the complexities of chemical behavior and reactivity in the main group elements. As a result, atoms and ions cannot be said to have exact sizes. in this section, we discuss how atomic and ion “sizes” are defined and obtained. A comparison of the dimensions of atoms or ions that have the same number of electrons but different nuclear charges, called an isoelectronic series, shows a clear correlation between increasing nuclear charge and decreasing size.
Electronegativity Atomic Size Missing Word An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. the higher the associated electronegativity, the more an atom or a substituent group attracts electrons. Understanding the trends in group properties such as atomic size, ionization energy, electronegativity, and electron affinity is fundamental to unraveling the complexities of chemical behavior and reactivity in the main group elements. As a result, atoms and ions cannot be said to have exact sizes. in this section, we discuss how atomic and ion “sizes” are defined and obtained. A comparison of the dimensions of atoms or ions that have the same number of electrons but different nuclear charges, called an isoelectronic series, shows a clear correlation between increasing nuclear charge and decreasing size.

Solution Bs Chemistry Short Qs About Trends Like Ionization Atomic As a result, atoms and ions cannot be said to have exact sizes. in this section, we discuss how atomic and ion “sizes” are defined and obtained. A comparison of the dimensions of atoms or ions that have the same number of electrons but different nuclear charges, called an isoelectronic series, shows a clear correlation between increasing nuclear charge and decreasing size.
Comments are closed.