Atomic Size Trend Ions
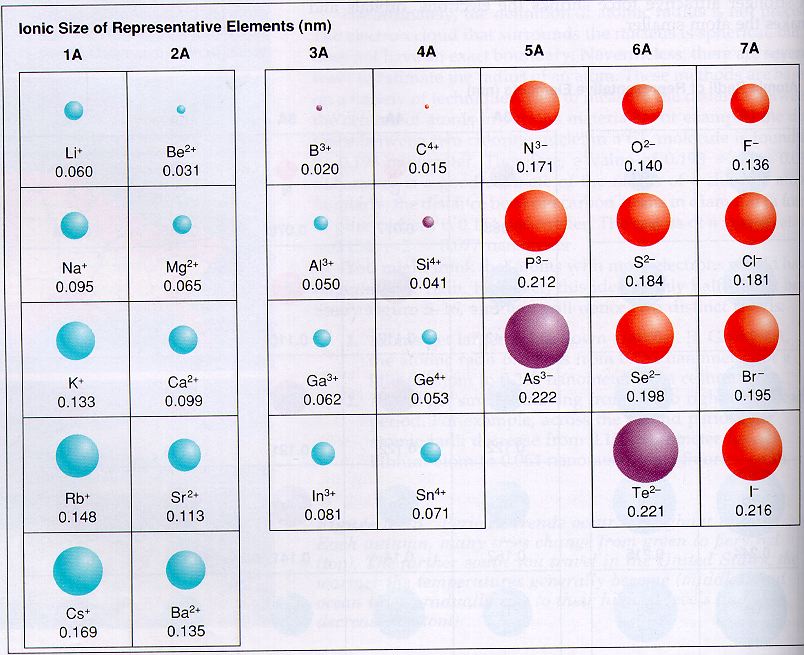
Atomic Size Trend Ions The ionic radii of cations and anions are always smaller or larger, respectively, than the parent atom due to changes in electron–electron repulsions, and the trends in ionic radius parallel those in atomic size. As a result, atoms and ions cannot be said to have exact sizes; however, some atoms are larger or smaller than others, and this influences their chemistry. in this chapter, we will discuss how atomic and ion “sizes” are defined and obtained.
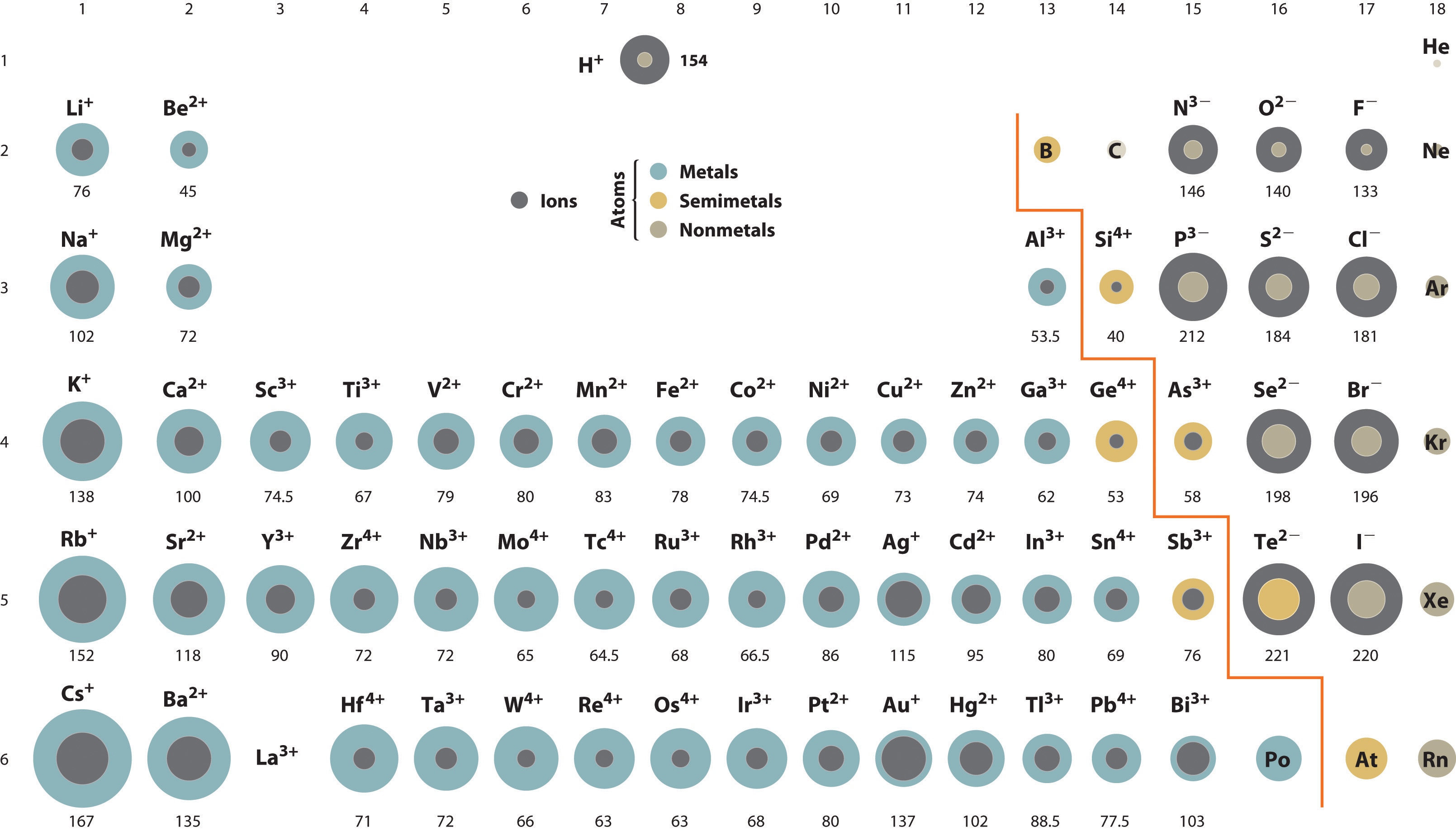
Atomic Size Trend Ions Explore the trend in atomic size across periods and groups. understand how electron configuration and effective nuclear charge influence atomic radius. A comparison of the dimensions of atoms or ions that have the same number of electrons but different nuclear charges, called an isoelectronic series, shows a clear correlation between increasing nuclear charge and decreasing size. The ionic radius trend indicates that ions become larger as you move down a group in the periodic table and smaller as you move across a period. Get the atomic radius and ionic radius definitions, learn the difference between them, and explore their periodic table trend.
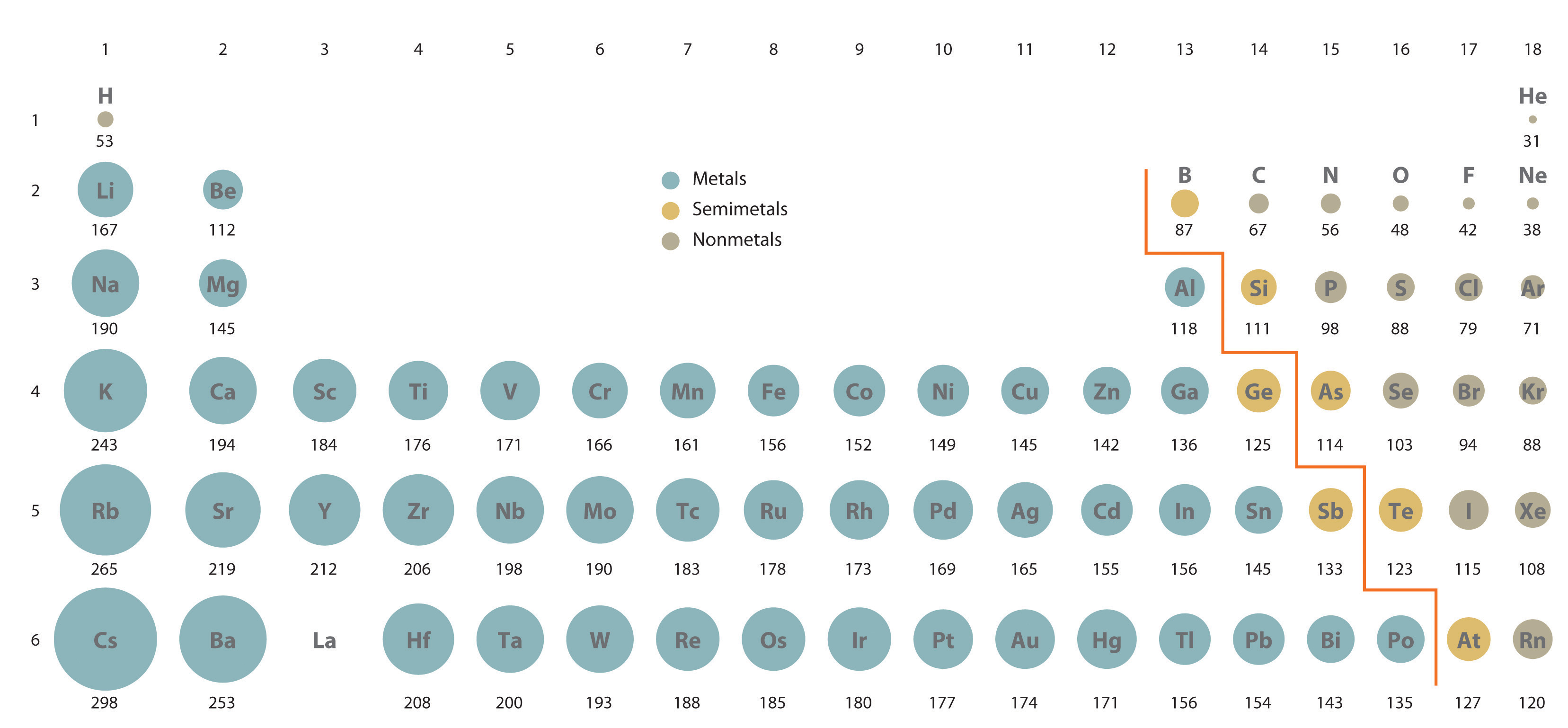
Atomic Size Trend Ions The ionic radius trend indicates that ions become larger as you move down a group in the periodic table and smaller as you move across a period. Get the atomic radius and ionic radius definitions, learn the difference between them, and explore their periodic table trend. With practice problems learn the periodic trends in the size of ions. learn why cations are smaller and anions are larger than neutral atoms. This page explores periodic trends of atomic properties, including atomic radius, ionization energy, and electron affinity, based on element positioning in the periodic table. Atomic radius is the distance between an atom’s nucleus and its outermost or valence electrons. on the other hand, the ionic radius is half the distance between two ions that barely touch each other in a compound. the atomic and ionic radii follow the same trend in the periodic table. Discover the ions size trend across periods and groups. learn how electron shells and nuclear charge affect ionic radii. click to explore key patterns and their impact on chemistry and materials science.
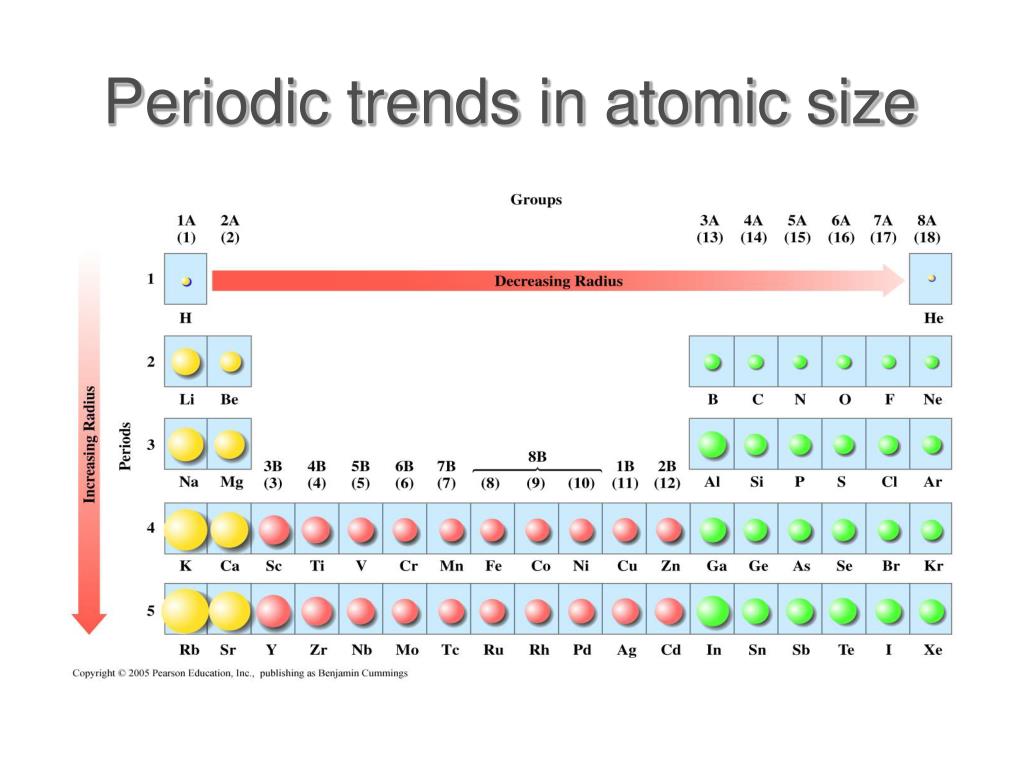
Ppt Why Periodic Powerpoint Presentation Free Download Id 1779615 With practice problems learn the periodic trends in the size of ions. learn why cations are smaller and anions are larger than neutral atoms. This page explores periodic trends of atomic properties, including atomic radius, ionization energy, and electron affinity, based on element positioning in the periodic table. Atomic radius is the distance between an atom’s nucleus and its outermost or valence electrons. on the other hand, the ionic radius is half the distance between two ions that barely touch each other in a compound. the atomic and ionic radii follow the same trend in the periodic table. Discover the ions size trend across periods and groups. learn how electron shells and nuclear charge affect ionic radii. click to explore key patterns and their impact on chemistry and materials science.

Periodic Trends Physical Atomic Ionic Radius Dp Ib Chemistry Hl Atomic radius is the distance between an atom’s nucleus and its outermost or valence electrons. on the other hand, the ionic radius is half the distance between two ions that barely touch each other in a compound. the atomic and ionic radii follow the same trend in the periodic table. Discover the ions size trend across periods and groups. learn how electron shells and nuclear charge affect ionic radii. click to explore key patterns and their impact on chemistry and materials science.
Comments are closed.