Ionic Radius Neet Lab
Ionic Radius Neetlab The ionic radius will be smaller than the atomic radius, depending on the electric charge of the ion. the ionic radius and the atomic radius are same for neutral atom. Neet 2026 preparation requires a strong understanding of ionic radius trends and question patterns ⚡in this short video, you will learn a quick trick to solv.
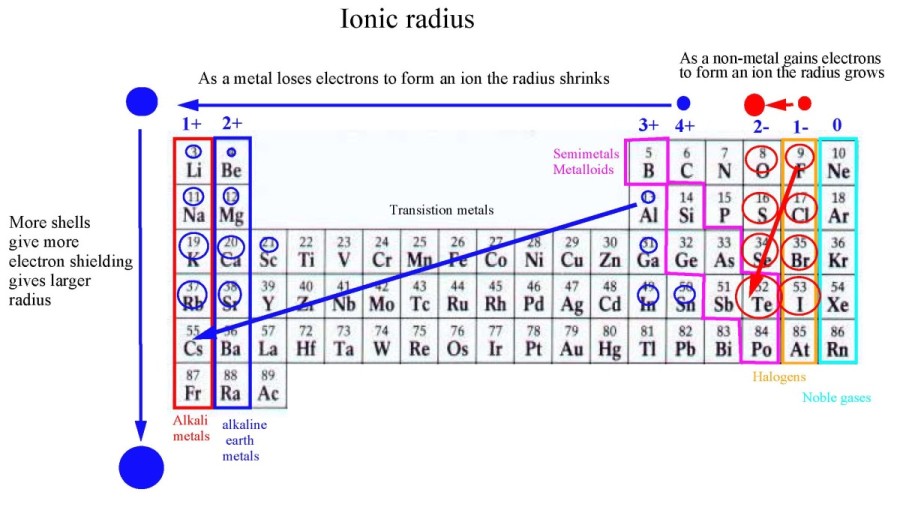
Ionic Radius Neetlab The ionic radius is defined as the distance between an ion’s nucleus and the ion’s outermost shell. in a periodic table, atoms add extra shells (number of electrons) as they move down a group, increasing the ionic radius of the elements. Ionic radius: ionic radius refers to the size of an ion. cations (positive ions) are smaller than their parent atoms, while anions (negative ions) are larger due to electron repulsion. In this video, we cover everything you need to know about ionic radius, along with a detailed explanation of classification of elements and periodicity in properties. Get atomic and ionic radii multiple choice questions (mcq quiz) with answers and detailed solutions. download these free atomic and ionic radii mcq quiz pdf and prepare for your upcoming exams like banking, ssc, railway, upsc, state psc.

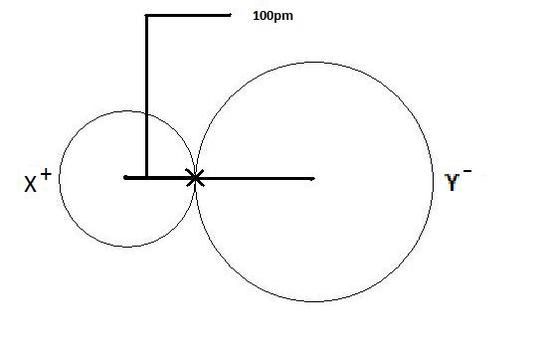
Ionic Radius Definition Examples Chart Periodic Trend 45 Off In this video, we cover everything you need to know about ionic radius, along with a detailed explanation of classification of elements and periodicity in properties. Get atomic and ionic radii multiple choice questions (mcq quiz) with answers and detailed solutions. download these free atomic and ionic radii mcq quiz pdf and prepare for your upcoming exams like banking, ssc, railway, upsc, state psc. Get the atomic radius and ionic radius definitions, learn the difference between them, and explore their periodic table trend. Understanding ionic radius is crucial in the study of chemistry, particularly when examining the behavior of ions. the ionic radius is defined as the distance from the nucleus of an ion to its outermost electron shell. The radius of a monatomic ion in an ionic crystal structure is called ionic radii. atomic and ionic radii are distances away from the nucleus or central atom, atomic radius is the distance away from the nucleus, and it increases as we go from top to bottom and decreases across the periodic table. The ionic radius can be described as the distance between the nucleus of an ion and the outermost shell of the ion. the atomic size of a cation will be smaller than that of the parent atom.
Comments are closed.