Atomic Vs Ionic Radius Why Ions Change Size

Ionic Radius Atomic Ionic Radius Trend Definition Differences The ionic radii of cations and anions are always smaller or larger, respectively, than the parent atom due to changes in electron–electron repulsions, and the trends in ionic radius parallel those in atomic size. Atomic radius is the size of a neutral atom; ionic radius is the size of an ion. when an atom becomes a cation (like na⁺), it gets smaller because it has fewer electrons and stronger pull per electron.

Atomic Radius And Ionic Radius While the atomic radius measures the size of a neutral atom, the ionic radius gauges the size of an electrically charged atom. the ionic radius is the radius of a monatomic ion of an element within an ionic crystal or half the distance between two bonded gas atoms. If the atom loses its outermost electron (positively charged or cation), the ionic radius is smaller than the atomic radius because the atom loses an electron energy shell. Learn about atomic and ionic radius for a level chemistry. find information on trends across periods and down groups of the periodic table. Ions may be larger or smaller than the neutral atom, depending on the ion's electric charge. when an atom loses an electron to form a cation, the other electrons are more attracted to the nucleus, and the radius of the ion gets smaller.

What Is An Ionic Radius At Leo Gatehouse Blog Learn about atomic and ionic radius for a level chemistry. find information on trends across periods and down groups of the periodic table. Ions may be larger or smaller than the neutral atom, depending on the ion's electric charge. when an atom loses an electron to form a cation, the other electrons are more attracted to the nucleus, and the radius of the ion gets smaller. Whether you choose to use van der waals radii or metallic radii as a measure of the atomic radius, for metals the ionic radius is smaller than either, so the problem doesn't exist to the same extent. When an atom forms an ion, it either gains electrons (anion) or loses electrons (cation). while forming an ion changes its size relative to the parent atom, the addition of a new, higher principal quantum number shell remains the dominant factor controlling size within the group. The atomic radius gives an estimation of the size of a neutral atom, while the ionic radius deals with the size of an atom that has gained or lost electrons, becoming an ion. When an atom gets an extra electron, it transforms into a negatively charged ion known as an anion, which has a larger ionic radius. when an atom loses an electron, it transforms into a positively charged ion known as a cation, which has a smaller ionic radius.

Ionic Radius Periodic Trends Whether you choose to use van der waals radii or metallic radii as a measure of the atomic radius, for metals the ionic radius is smaller than either, so the problem doesn't exist to the same extent. When an atom forms an ion, it either gains electrons (anion) or loses electrons (cation). while forming an ion changes its size relative to the parent atom, the addition of a new, higher principal quantum number shell remains the dominant factor controlling size within the group. The atomic radius gives an estimation of the size of a neutral atom, while the ionic radius deals with the size of an atom that has gained or lost electrons, becoming an ion. When an atom gets an extra electron, it transforms into a negatively charged ion known as an anion, which has a larger ionic radius. when an atom loses an electron, it transforms into a positively charged ion known as a cation, which has a smaller ionic radius.
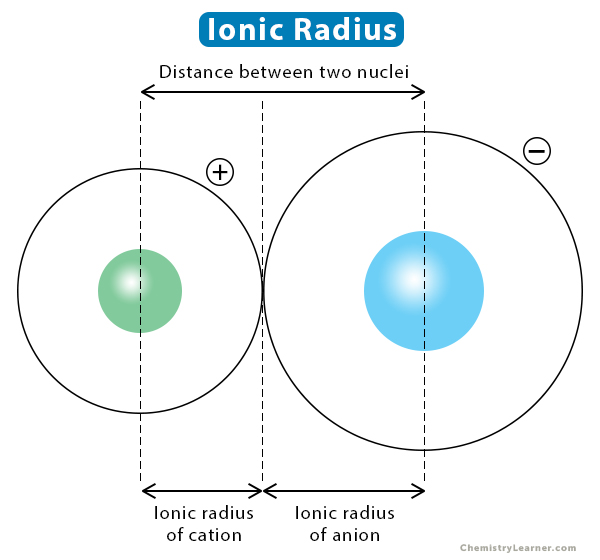
Ionic Radius Atomic Ionic Radius Trend Definition Differences The atomic radius gives an estimation of the size of a neutral atom, while the ionic radius deals with the size of an atom that has gained or lost electrons, becoming an ion. When an atom gets an extra electron, it transforms into a negatively charged ion known as an anion, which has a larger ionic radius. when an atom loses an electron, it transforms into a positively charged ion known as a cation, which has a smaller ionic radius.

Unit 4 The Periodic Table Ppt Download
Comments are closed.