Atomic Nucleus Size Comparison

Nucleus Size Comparison The Graph Compares Nucleus Size Of The dimensions of an electron are more complicated — they are so small that we cannot see them even with an electron microscope (in principle). they are believed to be smaller than 10—19 m, which is again roughly 100,000 times smaller than the nucleus of an atom. Compare the size of the nucleus with the size of the atom. so the atom is 10,000 times larger than the nucleus. another way of describing this fact is to say that the nucleus is 1 10,000 of.

Structure Of Atomic Nucleus And Its Size Science Vision The atom is about 100,000 times bigger than its nucleus. if the atomic nucleus were a pingpong ball (r ~ 1cm), its electrons would be in orbits about a kilometer away. In summary, the size of atoms, nuclei, and electrons underscores the gap between the microscopic and macroscopic scales. recognizing these vast differences aids in comprehending the fundamental principles of matter and the world around us. Understanding the relative sizes of atoms and their nuclei is essential for appreciating nuclear structure, modelling interactions, and interpreting experimental evidence about matter’s underlying organisation. An atom consists of a small, positively charged nucleus surrounded by electrons. the nucleus contains protons and neutrons; its diameter is about 100,000 times smaller than that of the atom.
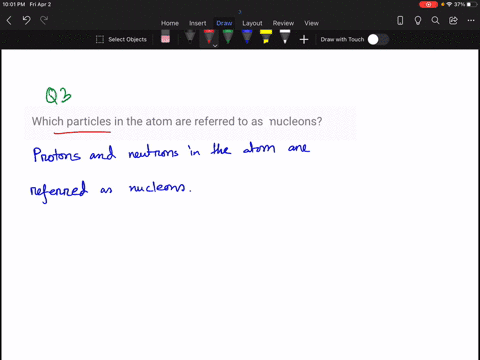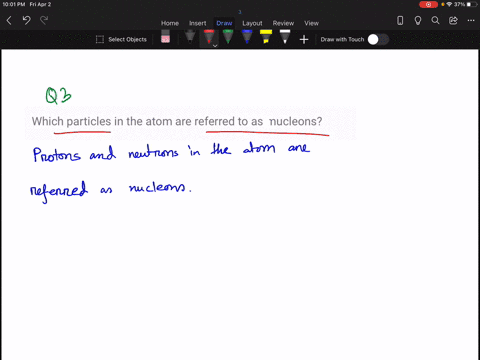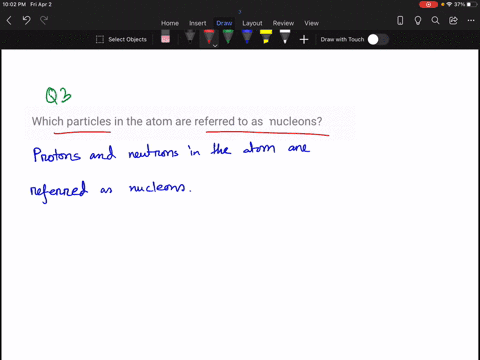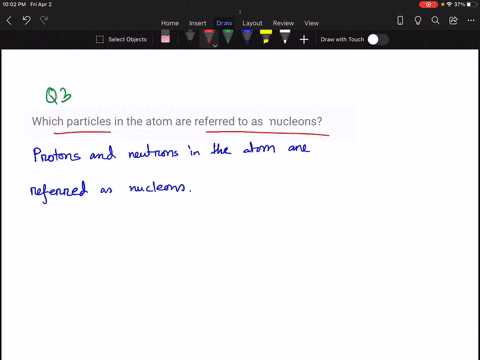
Solved How Large Is A Typical Atomic Nucleus And How Does The Size Of Understanding the relative sizes of atoms and their nuclei is essential for appreciating nuclear structure, modelling interactions, and interpreting experimental evidence about matter’s underlying organisation. An atom consists of a small, positively charged nucleus surrounded by electrons. the nucleus contains protons and neutrons; its diameter is about 100,000 times smaller than that of the atom. The size of an atomic nucleus is significantly smaller compared to the size of its electrons. the nucleus is the central core of an atom, consisting of protons and neutrons tightly bound together. the protons carry a positive charge, while the neutrons are electrically neutral. The nucleus is 40,000 times smaller than the atom. the relative size of the nucleus compared with an atom is that of the height of the pyramid of cheops (150m) to the radius of the earth (6,400,000m) (illustration m4). According to scattering experiments, the nucleus is spherical or ellipsoidal in shape, and about 1 100,000th the size of a hydrogen atom. if an atom were the size of a major league baseball stadium, the nucleus would be roughly the size of the baseball. The nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. a nucleus accounts for more than 99.9% of an atom’s mass but is 100,000 times smaller than it in size.

Illustration Of Chemistry Size Of The Elements Atomic Radius Atomic The size of an atomic nucleus is significantly smaller compared to the size of its electrons. the nucleus is the central core of an atom, consisting of protons and neutrons tightly bound together. the protons carry a positive charge, while the neutrons are electrically neutral. The nucleus is 40,000 times smaller than the atom. the relative size of the nucleus compared with an atom is that of the height of the pyramid of cheops (150m) to the radius of the earth (6,400,000m) (illustration m4). According to scattering experiments, the nucleus is spherical or ellipsoidal in shape, and about 1 100,000th the size of a hydrogen atom. if an atom were the size of a major league baseball stadium, the nucleus would be roughly the size of the baseball. The nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. a nucleus accounts for more than 99.9% of an atom’s mass but is 100,000 times smaller than it in size.
Comments are closed.