Atom Nucleus Size Comparison
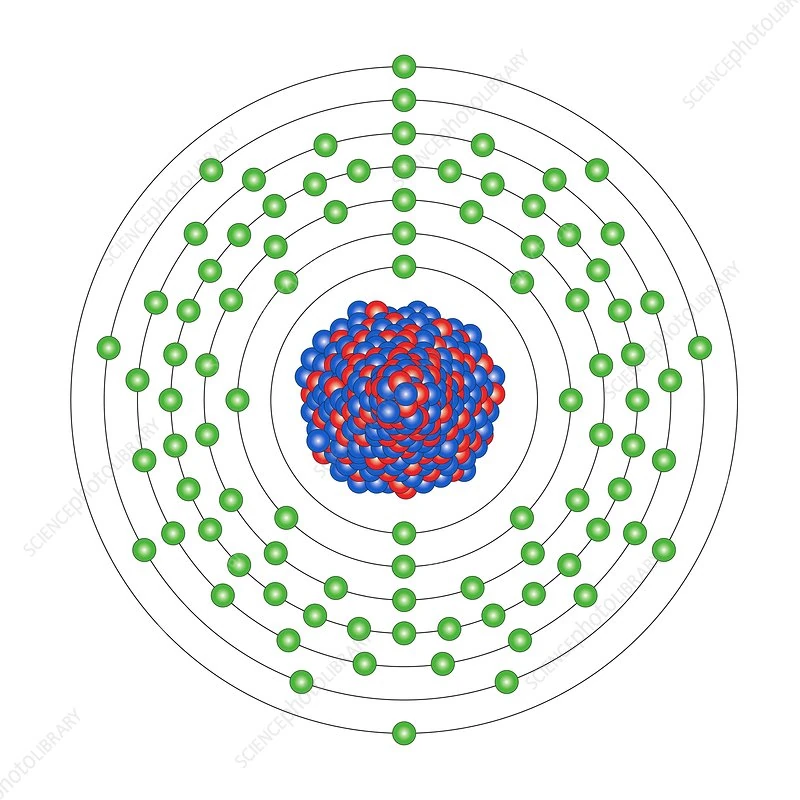
Uranium Nucleus Size Comparison Of Everything Wiki Fandom The dimensions of an electron are more complicated — they are so small that we cannot see them even with an electron microscope (in principle). they are believed to be smaller than 10—19 m, which is again roughly 100,000 times smaller than the nucleus of an atom. The atom is about 100,000 times bigger than its nucleus. if the atomic nucleus were a pingpong ball (r ~ 1cm), its electrons would be in orbits about a kilometer away.
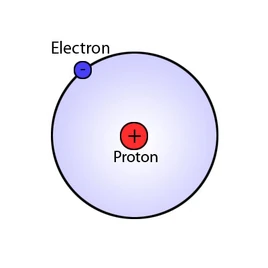
Hydrogen Nucleus Size Comparison Of Everything Wiki Fandom Compare the size of the nucleus with the size of the atom. so the atom is 10,000 times larger than the nucleus. another way of describing this fact is to say that the nucleus is 1 10,000 of the. An atom consists of a small, positively charged nucleus surrounded by electrons. the nucleus contains protons and neutrons; its diameter is about 100,000 times smaller than that of the atom. In summary, the size of atoms, nuclei, and electrons underscores the gap between the microscopic and macroscopic scales. recognizing these vast differences aids in comprehending the fundamental principles of matter and the world around us. Learn about relative sizes: atom and nucleus with ocr a level physics notes written by expert a level teachers. the best free online ocr a level resource trusted by students and schools globally.
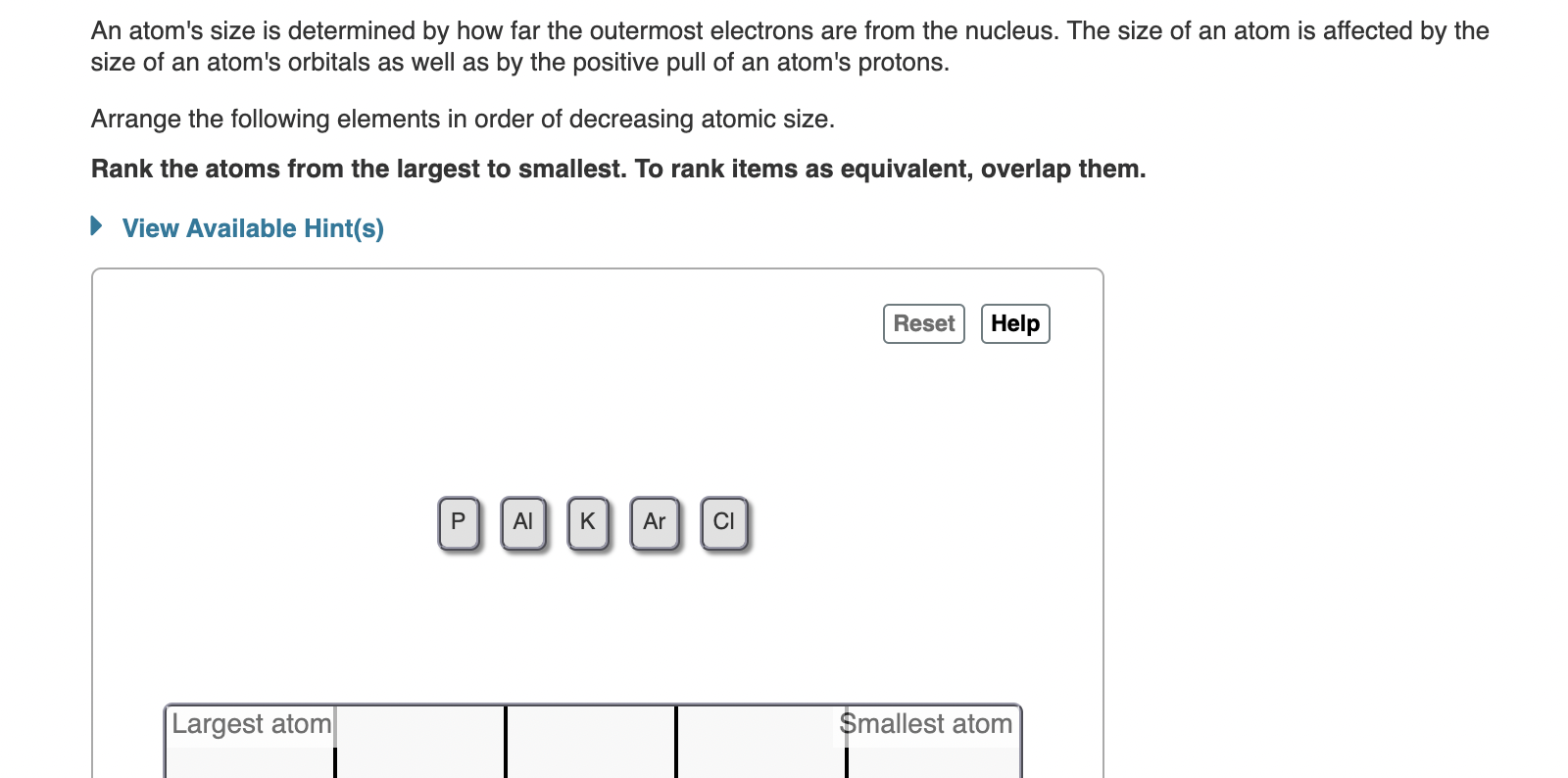
Solved An Atom S Size Is Determined By How Far The Outermost Chegg In summary, the size of atoms, nuclei, and electrons underscores the gap between the microscopic and macroscopic scales. recognizing these vast differences aids in comprehending the fundamental principles of matter and the world around us. Learn about relative sizes: atom and nucleus with ocr a level physics notes written by expert a level teachers. the best free online ocr a level resource trusted by students and schools globally. In other words, packing protons and neutrons in the nucleus gives approximately the same total size result as packing hard spheres of a constant size (like marbles) into a tight spherical or almost spherical bag (some stable nuclei are not quite spherical, but are known to be prolate). The nucleus is 40,000 times smaller than the atom. the relative size of the nucleus compared with an atom is that of the height of the pyramid of cheops (150m) to the radius of the earth (6,400,000m) (illustration m4). The size of an atomic nucleus is significantly smaller compared to the size of its electrons. the nucleus is the central core of an atom, consisting of protons and neutrons tightly bound together. the protons carry a positive charge, while the neutrons are electrically neutral. The nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. a nucleus accounts for more than 99.9% of an atom’s mass but is 100,000 times smaller than it in size.

Nucleus Of An Atom Atomic Nucleus Definition Structure Lesson In other words, packing protons and neutrons in the nucleus gives approximately the same total size result as packing hard spheres of a constant size (like marbles) into a tight spherical or almost spherical bag (some stable nuclei are not quite spherical, but are known to be prolate). The nucleus is 40,000 times smaller than the atom. the relative size of the nucleus compared with an atom is that of the height of the pyramid of cheops (150m) to the radius of the earth (6,400,000m) (illustration m4). The size of an atomic nucleus is significantly smaller compared to the size of its electrons. the nucleus is the central core of an atom, consisting of protons and neutrons tightly bound together. the protons carry a positive charge, while the neutrons are electrically neutral. The nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. a nucleus accounts for more than 99.9% of an atom’s mass but is 100,000 times smaller than it in size.

Atom Proton Neutron Nucleus Britannica The size of an atomic nucleus is significantly smaller compared to the size of its electrons. the nucleus is the central core of an atom, consisting of protons and neutrons tightly bound together. the protons carry a positive charge, while the neutrons are electrically neutral. The nucleus (plural, nuclei) is defined as the dense, central part of an atom, consisting of two subatomic particles, namely protons and neutrons. a nucleus accounts for more than 99.9% of an atom’s mass but is 100,000 times smaller than it in size.
Comments are closed.