Atom Nucleus Size

Atom Nucleus If we took a grain of poppy seed as an electron, the nucleus would be the size of a football field, so the entire atom would have a diameter of 10,000 km and an area roughly equivalent to the combined area of europe and africa. Protons and neutrons are bound together to form a nucleus by the nuclear force. the diameter of the nucleus is in the range of 1.70 fm (1.70 × 10−15 m[7]) for hydrogen (the diameter of a single proton) to about 11.7 fm for uranium. [8] .
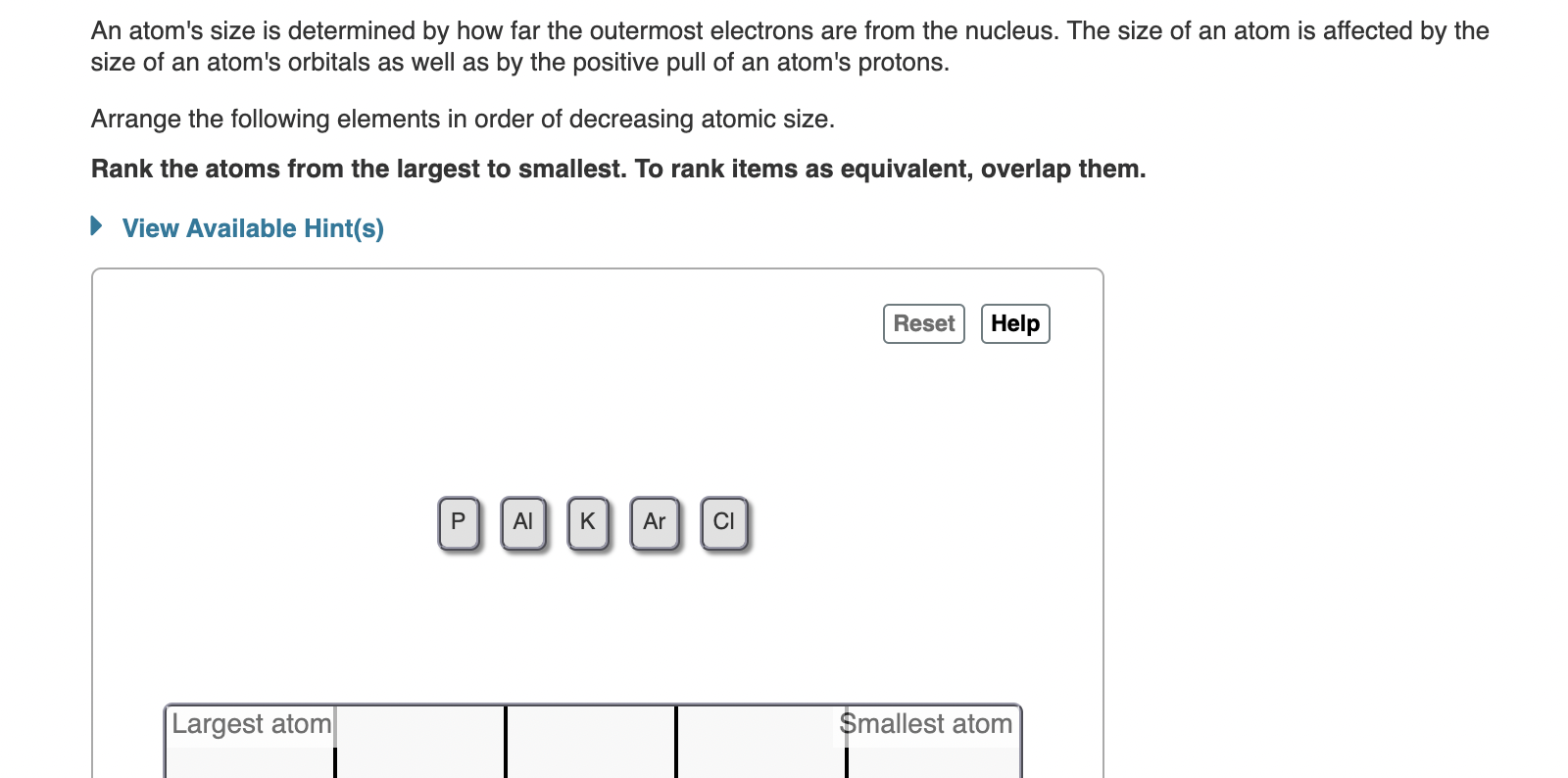
Solved An Atom S Size Is Determined By How Far The Outermost Chegg The nucleus is tiny when compared to the size of an atom. its diameter ranges between 1.6 fm (for the lightest atom, hydrogen) to about 15 fm (for the most massive atoms, such as uranium). this size amounts to only 1 100,000 times the radius of the whole atom. According to scattering experiments, the nucleus is spherical or ellipsoidal in shape, and about 1 100,000th the size of a hydrogen atom. if an atom were the size of a major league baseball stadium, the nucleus would be roughly the size of the baseball. An atom consists of a small, positively charged nucleus surrounded by electrons. the nucleus contains protons and neutrons; its diameter is about 100,000 times smaller than that of the atom. The nucleus of an atom is the very small dense region of an atom, in its center consisting of nucleons (protons and neutrons). the size (diameter) of the nucleus is in the range of 1.6 fm (10 15 m) (for a proton in light hydrogen) to about 15 fm (for the heaviest atoms, such as uranium).
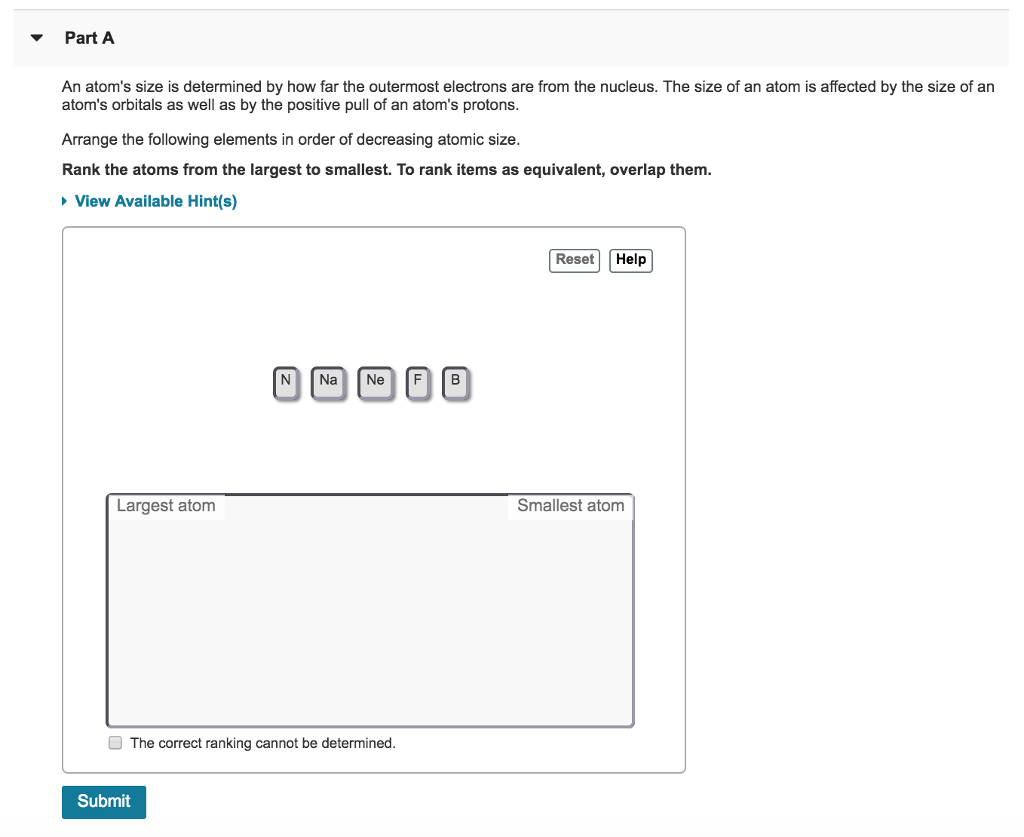
Solved Part A An Atom S Size Is Determined By How Far The Chegg An atom consists of a small, positively charged nucleus surrounded by electrons. the nucleus contains protons and neutrons; its diameter is about 100,000 times smaller than that of the atom. The nucleus of an atom is the very small dense region of an atom, in its center consisting of nucleons (protons and neutrons). the size (diameter) of the nucleus is in the range of 1.6 fm (10 15 m) (for a proton in light hydrogen) to about 15 fm (for the heaviest atoms, such as uranium). What are atoms made of? each atom consists of three types of particles: protons, neutrons and electrons. at the centre of an atom is a dense nucleus, which contains protons and neutrons, and is much smaller than the entire atom. if the nucleus of the atom were the size of a marble, the atom would be the size of a sports stadium. The size of an atomic nucleus is typically in the order of 10⁻¹⁵ metres (m), a unit also known as a femtometre (fm) or a fermi. the nuclear radius can range from approximately 1.75 fm for a simple nucleus like hydrogen to about 15 fm for heavier elements like uranium. Electrons have virtually no mass, but protons and neutrons have a lot of mass for their size. as a result, the nucleus has virtually all the mass of an atom. given its great mass and tiny size, the nucleus is very dense. The atomic model consists of a nucleus containing protons and neutrons, surrounded by electrons in shells. the numbers of particles in an atom can be calculated from its atomic number and.
Oxygen Atom Nucleus Structure Of The Hydrogen And Oxygen Atom What are atoms made of? each atom consists of three types of particles: protons, neutrons and electrons. at the centre of an atom is a dense nucleus, which contains protons and neutrons, and is much smaller than the entire atom. if the nucleus of the atom were the size of a marble, the atom would be the size of a sports stadium. The size of an atomic nucleus is typically in the order of 10⁻¹⁵ metres (m), a unit also known as a femtometre (fm) or a fermi. the nuclear radius can range from approximately 1.75 fm for a simple nucleus like hydrogen to about 15 fm for heavier elements like uranium. Electrons have virtually no mass, but protons and neutrons have a lot of mass for their size. as a result, the nucleus has virtually all the mass of an atom. given its great mass and tiny size, the nucleus is very dense. The atomic model consists of a nucleus containing protons and neutrons, surrounded by electrons in shells. the numbers of particles in an atom can be calculated from its atomic number and.

Oxygen Atom Nucleus Structure Of The Hydrogen And Oxygen Atom Electrons have virtually no mass, but protons and neutrons have a lot of mass for their size. as a result, the nucleus has virtually all the mass of an atom. given its great mass and tiny size, the nucleus is very dense. The atomic model consists of a nucleus containing protons and neutrons, surrounded by electrons in shells. the numbers of particles in an atom can be calculated from its atomic number and.
Comments are closed.