What Is Electron Affinity Eureka Blog
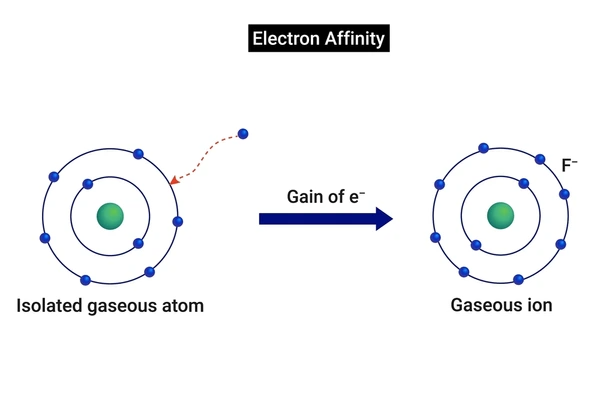
What Is Electron Affinity Eureka Blog Learn what electron affinity is, how it works, and why certain elements attract electrons more than others. The electron affinity (eea) of an atom or molecule is defined as the amount of energy released when an electron attaches to a neutral atom or molecule in the gaseous state to form an anion.
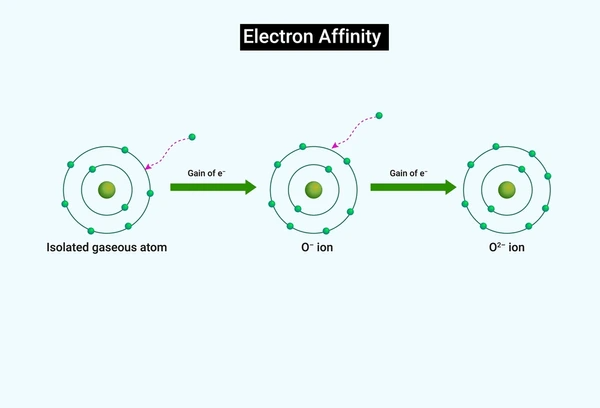
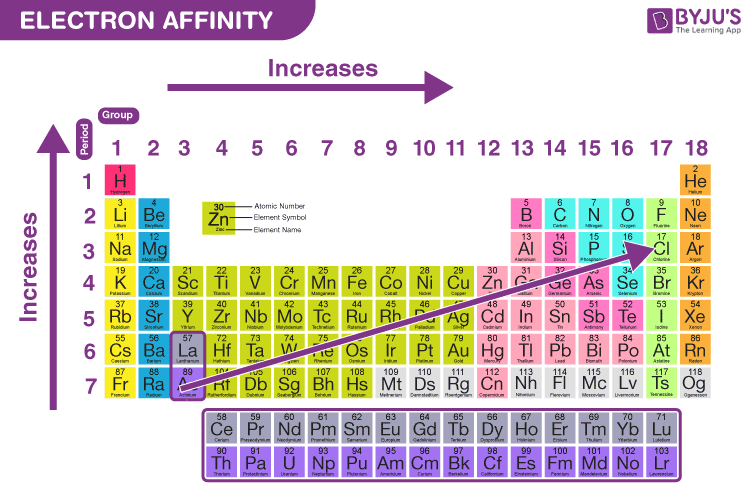
What Is Electron Affinity Eureka Blog Electron affinity is defined as the change in energy (in kj mole) of a neutral atom (in the gaseous phase) when an electron is added to the atom to form a negative ion. in other words, the neutral atom's likelihood of gaining an electron. The electron affinity is the difference of the energies of the system without an electron and that of the anion, e g e = ε 2. it is equal approximately to the negative energy of the virtual orbital on which the electron lands (if ε 2 <0 attaching an electron means energy lowering). This energy is expressed as the electron affinity, which is the energy released when an electron is attached to an atom of the element. in many cases, the electron affinity is positive, signifying that energy is indeed released when an electron attaches to an atom. Electron affinity is the energy change when a neutral gas phase atom gains an electron. for most elements, except noble gases, this is an exothermic process. electron affinity increases moving across a period and sometimes decreases moving down a group.
What Is Electron Affinity Eureka Blog This energy is expressed as the electron affinity, which is the energy released when an electron is attached to an atom of the element. in many cases, the electron affinity is positive, signifying that energy is indeed released when an electron attaches to an atom. Electron affinity is the energy change when a neutral gas phase atom gains an electron. for most elements, except noble gases, this is an exothermic process. electron affinity increases moving across a period and sometimes decreases moving down a group. Learn what electron affinity is, its definition, and how it varies across the periodic table. this guide explains the factors affecting electron affinity with simple examples, making it easy for students and chemistry learners to understand. What is electron affinity. learn its equation, examples, values, and change across a period & down a group in the periodic table. also, learn its exceptions. In this article, we will look into what electron affinity is, its periodic trends, the factors affecting electron affinity, and the values of electron affinity of different elements. Variation in electron affinities the electron affinity [ea] is the energy change for the process of adding an electron to a gaseous atom to form an anion (negative ion).

What Is Electron Affinity Eureka Blog Learn what electron affinity is, its definition, and how it varies across the periodic table. this guide explains the factors affecting electron affinity with simple examples, making it easy for students and chemistry learners to understand. What is electron affinity. learn its equation, examples, values, and change across a period & down a group in the periodic table. also, learn its exceptions. In this article, we will look into what electron affinity is, its periodic trends, the factors affecting electron affinity, and the values of electron affinity of different elements. Variation in electron affinities the electron affinity [ea] is the energy change for the process of adding an electron to a gaseous atom to form an anion (negative ion).
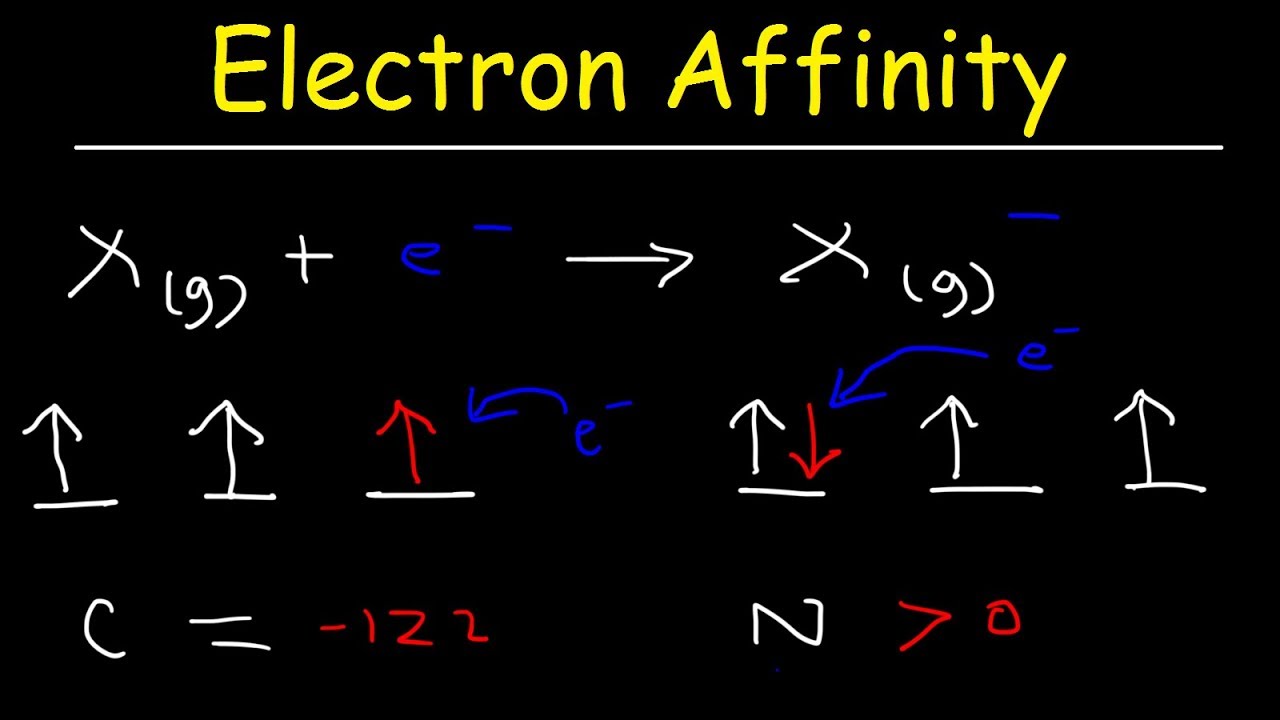
Hydrogen S Electron Affinity Explained Trends Uses More In this article, we will look into what electron affinity is, its periodic trends, the factors affecting electron affinity, and the values of electron affinity of different elements. Variation in electron affinities the electron affinity [ea] is the energy change for the process of adding an electron to a gaseous atom to form an anion (negative ion).
What Is Electron Affinity Definition Trends Equation With Videos
Comments are closed.