Supercritical Fluids

About Supercritical Fluids Supercritical Fluid Technologies A supercritical fluid (scf) is a substance at a temperature and pressure above its critical point, where distinct liquid and gas phases do not exist, but below the pressure required to compress it into a solid. [1]. The journal of supercritical fluids publishes original and review articles on the science and technology of supercritical fluids and processes. it covers topics such as thermodynamics, kinetics, properties, separations, applications, sustainability, and engineering aspects of supercritical fluids.
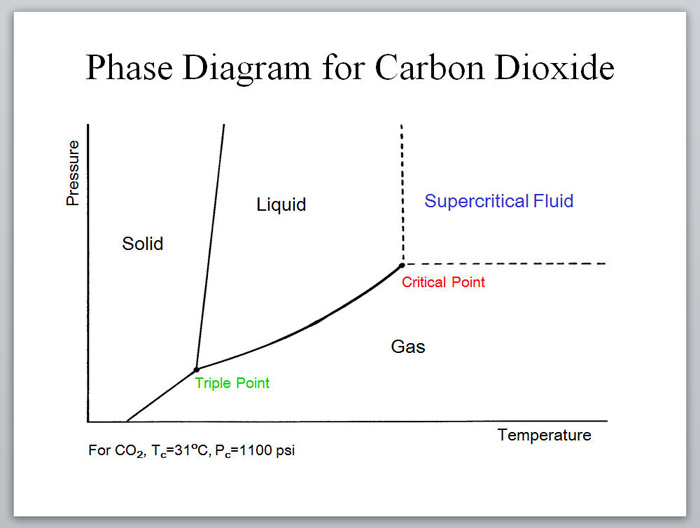
About Supercritical Fluids Supercritical Fluid Learn what supercritical fluids are, how they differ from liquids and gases, and what are their properties and phase diagrams. explore the examples of supercritical fluids such as co 2 and water, and their applications in extraction, chromatography, and sterilization. A supercritical fluid is a species held at a temperature and a pressure that exceeds its critical point. under these conditions the species is neither a gas nor a liquid. This review provides an overview of the basic principles of supercritical fluids, their unique properties, and their recent advances and applications in different scientific fields. Learn about supercritical fluids, their unique characteristics, and their uses in various fields. this review article covers the basic principles, the phase transition, the density and compressibility, and the solvating power of supercritical fluids.

How Do Supercritical Fluids Work Supercritical Fluid Technologies This review provides an overview of the basic principles of supercritical fluids, their unique properties, and their recent advances and applications in different scientific fields. Learn about supercritical fluids, their unique characteristics, and their uses in various fields. this review article covers the basic principles, the phase transition, the density and compressibility, and the solvating power of supercritical fluids. A supercritical fluid is a substance that exists at a temperature and pressure beyond its specific critical point, where the distinct boundaries between liquid and gas phases cease to exist. this state of matter is a hybrid—not quite a liquid and not quite a gas—that possesses unique properties. A supercritical fluid (scf) is a highly compressed fluid that exhibits the combined properties of gases and liquids. scientists create scfs by increasing both the temperature and pressure of a substance beyond its critical point. Learn about supercritical fluids, a state of matter that has no definite phase and exhibits properties of both gases and liquids. find out how to create, identify and use supercritical fluids, and see examples of common substances with critical points. Cagniard de la tour showed in 1822 that there is a critical temperature above which a single substance can only exist as a fluid and not as either a liquid or gas. he heated substances, present as both liquid and vapor, in a sealed cannon, which he rocked back and.
Comments are closed.