Supercritical Fluids Explained

About Supercritical Fluids Supercritical Fluid Technologies A supercritical fluid (scf) is a substance at a temperature and pressure above its critical point, where distinct liquid and gas phases do not exist, but below the pressure required to compress it into a solid. [1]. What is a supercritical fluid explained with phase diagrams. also, learn its examples, properties, advantages, and applications.

How Do Supercritical Fluids Work Supercritical Fluid Technologies A supercritical fluid is a fluid at a temperature and pressure above its critical point, where liquid and gas phases cannot be distinguished. it can dissolve materials like a liquid, and diffuse through solids like a gas. Explore the fascinating hybrid state of matter known as a supercritical fluid, understanding its characteristics and diverse real world applications. Supercritical fluids, such as supercritical water and carbon dioxide, have found applications in various green chemistry processes, including supercritical water oxidation and carbon dioxide utilization. A supercritical fluid is a substance that exists at a temperature and pressure beyond its specific critical point, where the distinct boundaries between liquid and gas phases cease to exist. this state of matter is a hybrid—not quite a liquid and not quite a gas—that possesses unique properties.
Supercritical Fluids Supercritical fluids, such as supercritical water and carbon dioxide, have found applications in various green chemistry processes, including supercritical water oxidation and carbon dioxide utilization. A supercritical fluid is a substance that exists at a temperature and pressure beyond its specific critical point, where the distinct boundaries between liquid and gas phases cease to exist. this state of matter is a hybrid—not quite a liquid and not quite a gas—that possesses unique properties. Learn what a supercritical fluid is, how it behaves, and why it's important in modern extraction, chromatography, and analytical chemistry applications. This review provides an overview of the basic principles of supercritical fluids, their unique properties, and their recent advances and applications in different scientific fields. A supercritical fluid is any substance at a temperature and pressure above its thermodynamic critical point. it has the unique ability to diffuse through solids like a gas, and dissolve materials like a liquid. Explore the unique properties and applications of supercritical fluids, particularly co2, in extraction processes, focusing on eco friendly and efficient techniques.
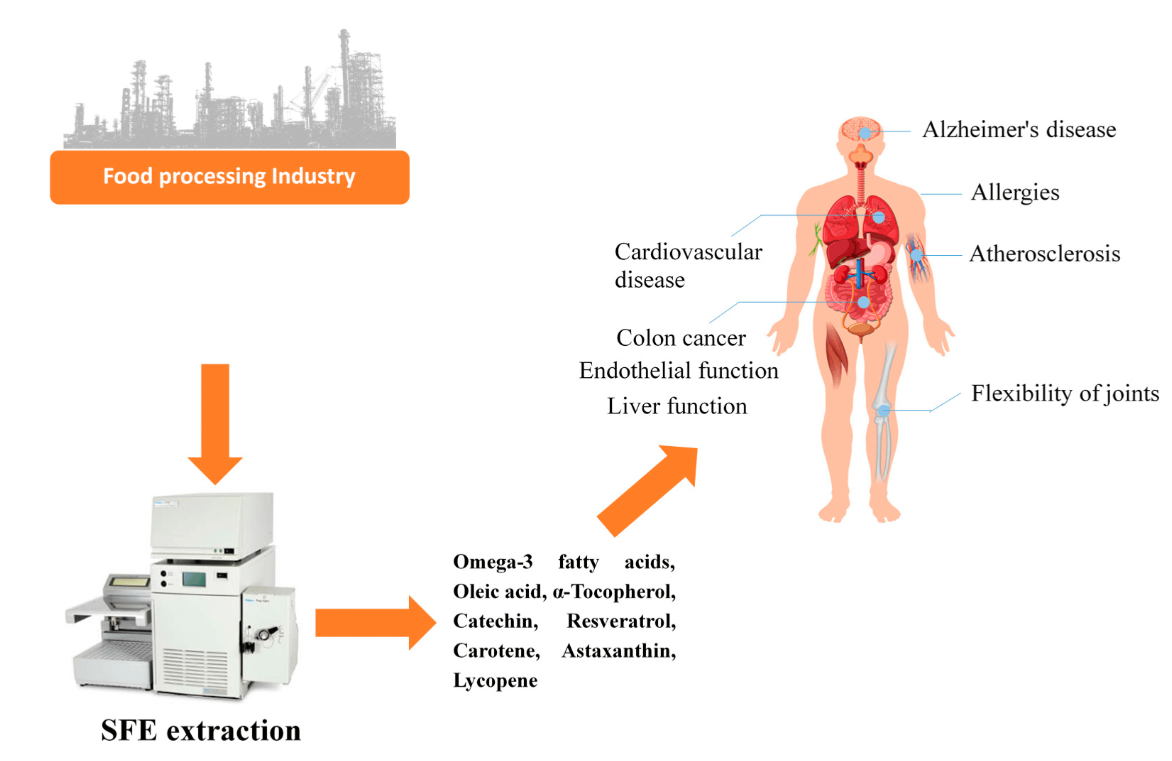
Supercritical Fluids Rayen Ghanmi Learn what a supercritical fluid is, how it behaves, and why it's important in modern extraction, chromatography, and analytical chemistry applications. This review provides an overview of the basic principles of supercritical fluids, their unique properties, and their recent advances and applications in different scientific fields. A supercritical fluid is any substance at a temperature and pressure above its thermodynamic critical point. it has the unique ability to diffuse through solids like a gas, and dissolve materials like a liquid. Explore the unique properties and applications of supercritical fluids, particularly co2, in extraction processes, focusing on eco friendly and efficient techniques.
Comments are closed.