Supercritical Carbon Dioxide Term

Supercritical Carbon Dioxide Cycles For Generation Iv Nuclear Reactors Supercritical carbon dioxide (sco 2[disputed – discuss]) is a fluid state of carbon dioxide where it is held at or above its critical temperature and critical pressure. Super critical carbon dioxide (scco2) is a modified steam explosion involving co 2 instead of atmospheric air. a specified amount of biomass is wetted and loaded in a reactor after which the pressure within the reactor is built up by means of super critical co 2.
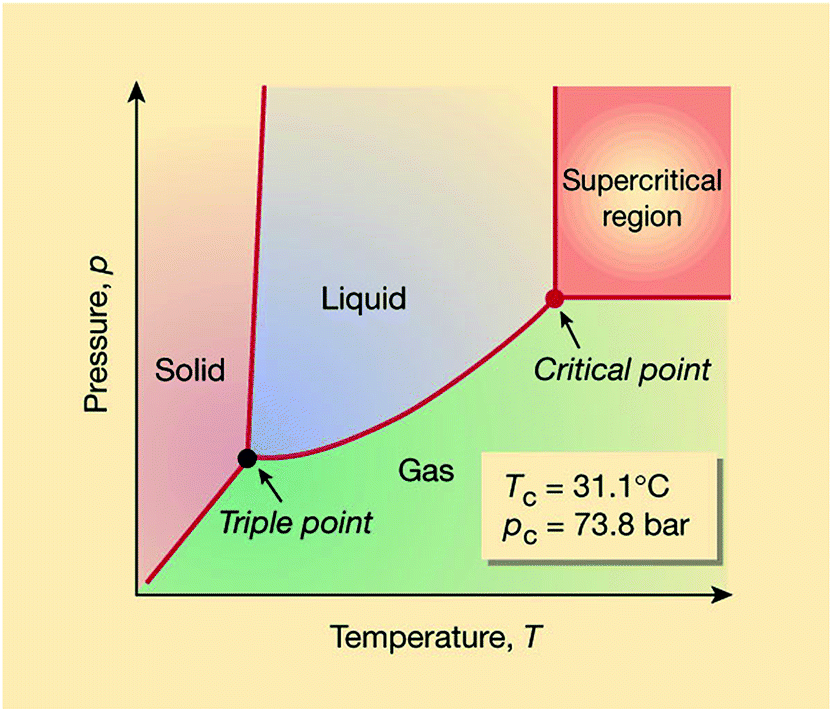
Schematic Phase Diagram For Supercritical Carbon Dioxide Download Supercritical carbon dioxide is a state of carbon dioxide where it exhibits properties between those of a gas and a liquid, achieved at specific temperature and pressure conditions. Supercritical co 2 is carbon dioxide held at or above its critical temperature (31.1°c) and critical pressure (73.8 bar), where it behaves as both a gas and a liquid. in this state, it has high solvating power and is used in processes like decaffeination, extraction, and cleaning. Supercritical carbon dioxide (scco₂) is a state of carbon dioxide that occurs when it is heated and pressurized beyond its critical point, resulting in properties that are intermediate between those of a gas and a liquid. Carbon dioxide usually behaves as a gas in air or as a solid called dry ice when frozen. above its critical temperature and pressure, it behaves like a supercritical fluid and can adopt properties midway between a gas and a liquid.
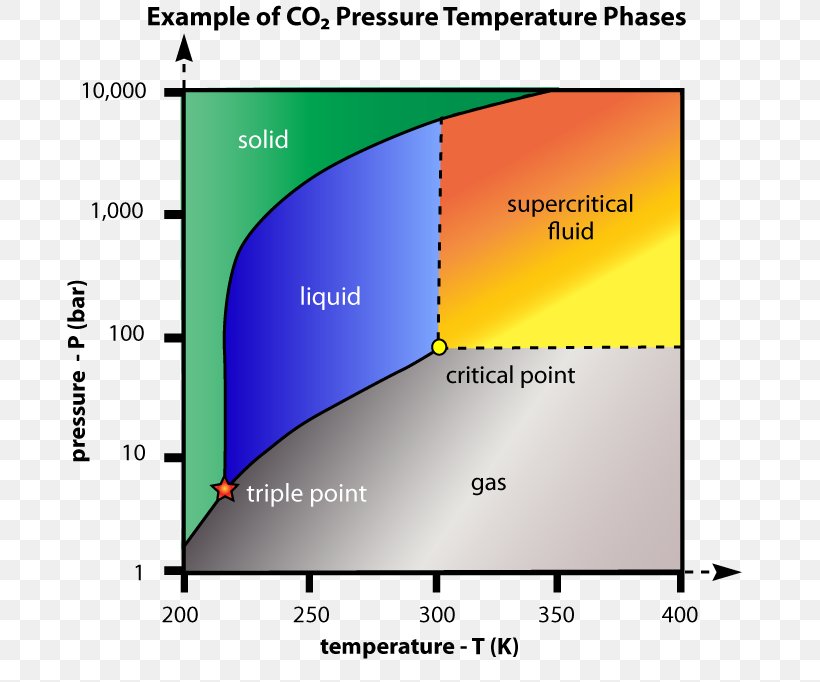
Critical Point Supercritical Fluid Supercritical Carbon Dioxide Phase Supercritical carbon dioxide (scco₂) is a state of carbon dioxide that occurs when it is heated and pressurized beyond its critical point, resulting in properties that are intermediate between those of a gas and a liquid. Carbon dioxide usually behaves as a gas in air or as a solid called dry ice when frozen. above its critical temperature and pressure, it behaves like a supercritical fluid and can adopt properties midway between a gas and a liquid. Supercritical carbon dioxide (scco 2) could be one aspect of a significant and necessary movement towards green chemistry, being a potential replacement for volatile organic compounds (vocs). unfortunately, carbon dioxide has a notoriously poor solubilising power and is famously difficult to handle. However, there exists another phase of co2 known as the supercritical state. this state is achieved when co2 surpasses its critical point, where specific pressure and temperature conditions allow for the coexistence of gas and liquid phases. Supercritical co2 is a state where carbon dioxide exhibits the properties of both a liquid and a gas, making it a powerful and selective solvent. the applications of supercritical co2 connect directly to a more sustainable lifestyle, often in unseen ways. What does supercritical carbon dioxide mean? supercritical carbon dioxide (co 2) is a fluid state of co 2 where it is heated and held at or above its critical temperature and pressure. in this supercritical phase, co 2 exhibits properties and behaviors between that of a liquid and a gas.
Supercritical Co2 Co2 三相図 Experimental Investigation Of Supercritical carbon dioxide (scco 2) could be one aspect of a significant and necessary movement towards green chemistry, being a potential replacement for volatile organic compounds (vocs). unfortunately, carbon dioxide has a notoriously poor solubilising power and is famously difficult to handle. However, there exists another phase of co2 known as the supercritical state. this state is achieved when co2 surpasses its critical point, where specific pressure and temperature conditions allow for the coexistence of gas and liquid phases. Supercritical co2 is a state where carbon dioxide exhibits the properties of both a liquid and a gas, making it a powerful and selective solvent. the applications of supercritical co2 connect directly to a more sustainable lifestyle, often in unseen ways. What does supercritical carbon dioxide mean? supercritical carbon dioxide (co 2) is a fluid state of co 2 where it is heated and held at or above its critical temperature and pressure. in this supercritical phase, co 2 exhibits properties and behaviors between that of a liquid and a gas.

Supercritical Carbon Dioxide Term Supercritical co2 is a state where carbon dioxide exhibits the properties of both a liquid and a gas, making it a powerful and selective solvent. the applications of supercritical co2 connect directly to a more sustainable lifestyle, often in unseen ways. What does supercritical carbon dioxide mean? supercritical carbon dioxide (co 2) is a fluid state of co 2 where it is heated and held at or above its critical temperature and pressure. in this supercritical phase, co 2 exhibits properties and behaviors between that of a liquid and a gas.

Ppt What Is Green Chemistry Powerpoint Presentation Free Download
Comments are closed.